Current Concept Review
Lateral Epiphyseal Narrowings with Absent Fibula Conform to a Vascular Pattern Deficiency
1Department of Orthopedic Surgery, SUNY Upstate Medical University, Syracuse, NY; 2Department of Cell and Developmental Biology, SUNY Upstate Medical University, Syracuse, NY; 3Alan and Marlene Norton College of Medicine, SUNY Upstate Medical University, Syracuse, NY; 4Department of Pediatrics, SUNY Upstate Medical University, Syracuse, NY
Correspondence: Jason A. Horton, PhD; Department of Orthopedic Surgery, SUNY Upstate Medical University, 3119 Institute for Human Performance, Syracuse, NY 13210. E-mail: [email protected]
Received: June 3, 2022; Accepted: June 17, 2022; Published: August 1, 2022
DOI: 10.55275/JPOSNA-2022-0083
Volume 4, Number 3, August 2022
Abstract:
Lateral epiphyseal narrowing with valgus deformities of the knee and ankle are cardinal radiographic features of Congenitally Shortened Limbs (CSL) exhibiting Congenital Fibular Deficiency (CFD). Radiographs of typical CFD limbs reveal valgus deformities and lateral epiphyseal reductions at both the knee and ankle. Arteriography of CSL reveals not only absence of adult vessels, but preservation of primitive vessels. Specifically, the superior and inferior lateral genicular arteries that habitually serve the lateral femoral condyle (LFC) and the lateral aspect of the proximal tibial epiphysis are absent in CSL. Similarly, the usual arteries supplying the distal tibial epiphysis are absent, except for a singular periosteal vessel, which independently serves the anteromedial portion.
Active transitioning of the single primitive Axial Artery (AA) to the usual adult pattern of the lower limb accompanies periods of rapid growth and ossification of the anlagen. Dysgenesis of the lateral arteries supplying the secondary ossification centers leads to asymmetric growth of the developing epiphyses, caused by relative medial overgrowth. Since vascular dysgenesis manifests at sites of failed embryonic transition of the proximal Femur, fibula, and midline Metatarsals (FfM) in CSL, it is suspected that vascular dysgenesis also underlies the impaired growth of the reduced epiphyses as seen in CSL. This paper reviews these concepts.
Key Concepts:
- Undergrowth of the lateral epiphyses leads to valgus deformities of both the knee and ankle in CSL.
- Undergrowth is due to embryonic dysgenesis of lateral vasculature at and below the knee.
- In CSL, some adult vessels fail to form while more primitive vessels are preserved.
- Valgus angular deformities in CSL result from lateral reductions of the secondary ossification centers due to vascular dysgeneses that occurred before the epiphyses formed.
| Abbreviations CSL = Congenitally Shortened Limb FfM = Syndrome of Proximal Femur, Fibula, and Midline Metatarsals CFD = Congenital Fibular Deficiency LFC = Lateral Femoral Condyle PFFD = Proximal Femoral Focal Deficiency FA = Femoral Artery ATA = Anterior Tibial Artery PTA = Posterior Tibial Artery PA = Peroneal Artery MPA = Medial Plantar Artery AA = Axial Artery CR = Crown Rump Length of Embryo |
Introduction
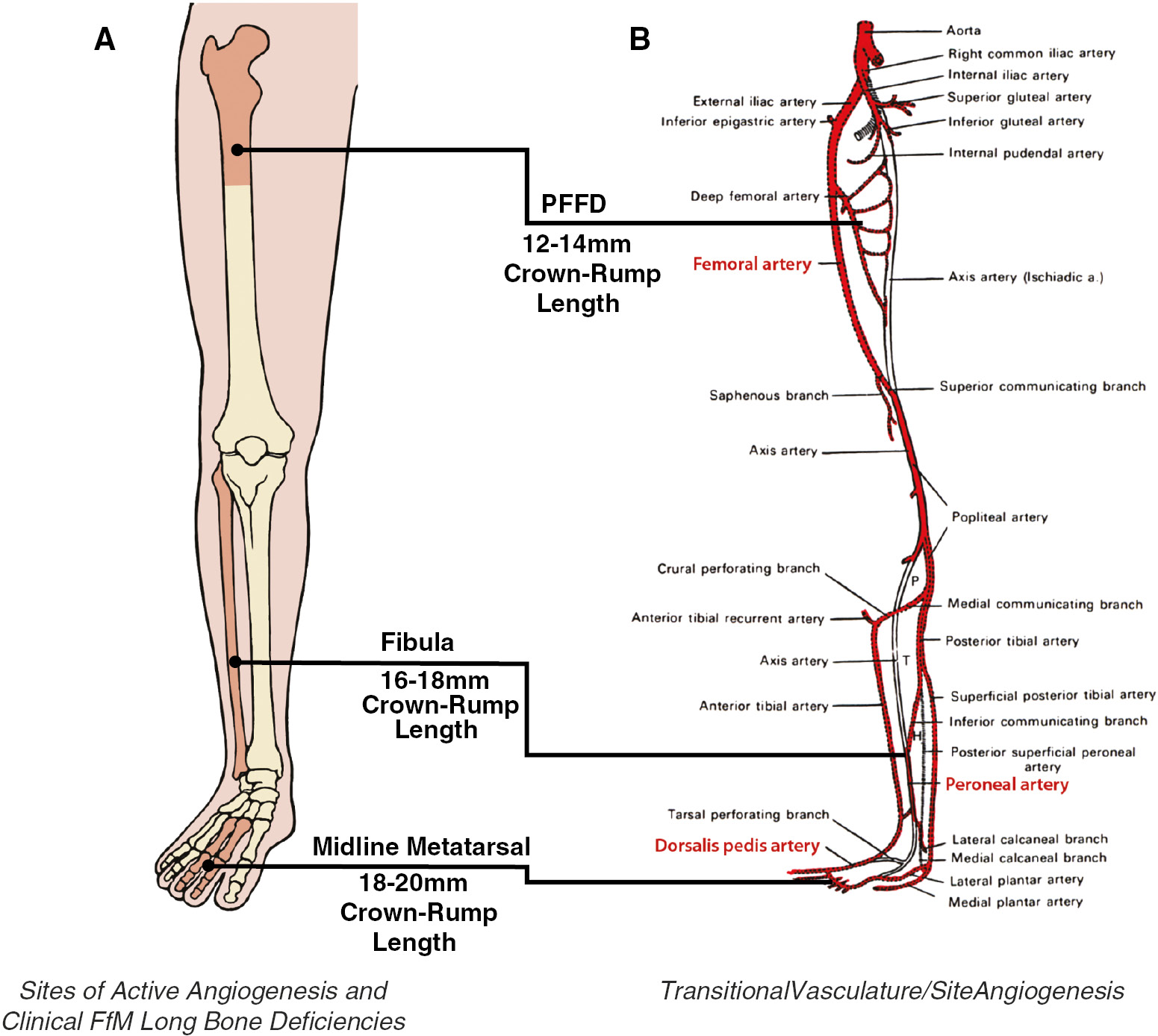
Radiographically absent fibula is the most frequent human congenital long bone deficiency of Congenitally Shortened Limbs (CSL).1 Arnold2 and others3–6 observed that equinovalgus deformities of the ankle/foot are part of a variable spectrum of anomalies involving the entire CSL, which encompasses the ipsilateral hip, femur, knee, tibia/fibula, ankle, and foot. Valgus deformities at the knee7 and ankle2,8 are cardinal findings of the CFD limb.4–6 Radiographic absence and/or delay of ossification of the long bones are regular features of the entire CSL, particularly of the proximal femur, fibula, and metatarsals (Figure 1).2,9,10
Figure 1. A) Congenital long bone deficiencies of the human lower limb cluster at three distinct skeletal locations: the proximal femur in PFFD, the fibula in CFD, and the midline metatarsals. B) Illustration of the stages of arterial development in the lower limb to the adult form Crown-rump (CR) length indicates the developmental stage and at which each bony segment forms,11,12 and the respective supplying arterial branch transitions.13,14 This shared chronology implies that vascular insults at or prior to ossification result in the bony dystrophism. Reproduced from Senior, with modification.13,15 These long bone dystrophisms occur at progressively more distal sites, corresponding to the times and locations where vascular transitioning and angiogenesis occur during the sixth and seventh weeks of embryonic development. P indicates the position of the popliteus, T indicates the tibialis posterior, and H indicates the flexor hallucis longus.
Such bony dysmorphisms have been attributed to acquired post-specification developmental errors.16,17 The developmental cause(s) of the full spectrum of CSL deficiencies are incompletely understood. In the absence of other genetic anomalies, such dysmorphisms are likely the result of deviations from otherwise normal developmental processes.18
The most severe long bone reductions in CSL manifest at three noncontiguous locations: the proximal femur (PFFD),19 the fibula (CFD),2–6,19,20 and the midline metatarsals (M) (Figure 1A).20,21 This triad of dystrophisms syndromically corresponds to progressively later and more distal sites of evolving angiogenesis during transition from the primitive embryonic Axial Artery (AA) of the early limb bud to the more familiar and complex pattern of the adult limb (Figure 1B).13–16 Each dystrophic long bone segment conforms to a common theme of failed transitioning of the primitive axial limb artery transitions to the adult pattern at each segment (Table 1).13–16
Table 1. Spatial-Temporal Sequence of Bone and Arterial Development in the Lower Limb
| Skeletal Dysmorphism | Forming Artery | Crown-Rump Length |
|---|---|---|
| PFFD | Femoral a. | 12-14 mm22 |
| CFD | Peroneal a. | 16-18 mm23 |
| Midline Metatarsal | Dorsalis Pedis & Medial Plantar aa. | 18-20 mm24 |
Valgus angulations at the knee and ankle in CSL have been attributed to unspecified hypoplasia of the lateral compartments of the limb, epiphyseal undergrowth, and/or mechanical tethering.7,8,25–28 Radiography of CFD limbs characteristically reveals that both the proximal (Figure 2) and distal (Figures 3 & 4) tibial bony epiphyses are smaller laterally and are irregular.2
Figure 2. A) Comparative standing anteroposterior radiograph of a normal right and CFD-affected left limb of a juvenile patient. Reductions of the lateral femoral condyle and the lateral portion of the proximal tibial epiphysis are evident; the fibula is radiographically absent. B) A graphic illustration of the radiographically diminished (gray) and absent skeletal structures (dashed) in the CFD limb, superimposed over the contralateral (solid line), demonstrating an absent fibula as well as reduction of the lateral femoral condyle and proximal tibial epiphysis.

Figure 3. Comparative anteroposterior radiographs of the ankles of a juvenile CFD patient. A) The CFD affected ankle demonstrates a triangular distal tibial epiphysis with the apex narrowed toward the midline; like the ipsilateral fibula, the lateral aspect of the epiphysis is radiolucent. B) Radiograph of the contralateral ankle of the same patient displays a normal, trapezoidal distal tibial epiphysis. C) Illustrative superimposition of images A & B, highlighting the normal tibia (shaded) and radiographic absence of the fibula and lateral bony epiphyseal structures (dashed) in the CFD limb.
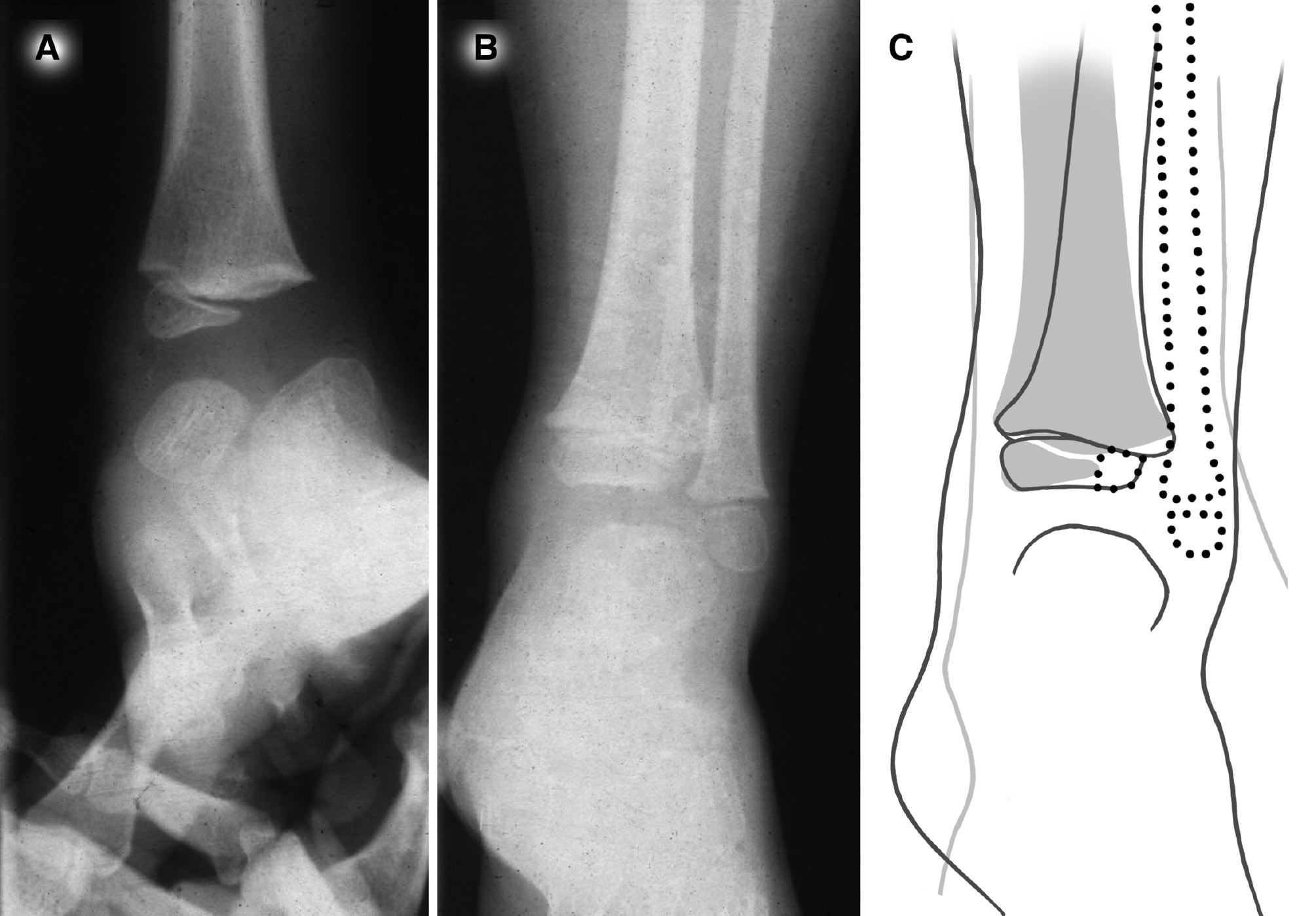
Figure 4. A) Antero-posterior and B) lateral radiographs of a juvenile CFD limb reveals anterior bowing of the tibial shaft, radiolucent fibular anlage, and deficiency of the posterior distal tibial epiphysis, which tapers posteriorly.23 C) Illustration of B, highlighting the structures that are present (solid line) and approximate location of the deficient structures CFD (dashed lines). D, E) Anteroposterior and F, G) lateral radiographs with corresponding illustrations of the same limb shown in A-C after skeletal maturity.30 The fibular anlage has mineralized and is now radiographically apparent. The distal tibial articulation faces posteriorly and laterally to articulate with the flattened head of the deformed talus. Illustrations E and G outline the missing posterior and lateral portions of the distal tibial epiphysis (dashed) of the articular surface of the distal tibia at the plafond, which extend to the visible physeal scars (white arrows). Images reproduced by permission.

Hypoplasia of the LFC is the single most frequent finding (93%) in all classes of CFD limbs.4,6 Furthermore, the CFD limb displays severe deficiencies of the distal tibial plafond, which slopes almost vertically from the midline, facing laterally and posteriorly (Figure 4).8,23,29 The consequence of these arterial deficiencies are observed clinically as dysmorphism and delayed ossification of the LFC and the lateral portions of the proximal (Figure 2) and distal tibial epiphyses (Figure 3) and the resulting valgus deformities. This Current Concept Review explores the anatomic and temporal relationship of defects in embryonic vascular development and transitioning to the lateral epiphyseal reductions and associated valgus deformities encountered in the clinic.
Normal Embryonic-to-Adult Transitions of the Lower Limb Arteries
The vascular system is the first tissue to differentiate from the embryonic mesoderm through a process termed vasculogenesis. It commences as a syncytium of interconnecting vascular channels which are further modified by differentiation, growth, and migration. The earliest primitive limb arteries form at discrete locations, coupled to the specification, patterning, and subsequent growth of the embryonic tissues they will serve. Patterning of the embryonic vessels is not an intrinsic property of arteries themselves. Rather, the arterial endothelium remodels by enlarging, branching, converging, or receding in response to instructive signals from their client tissues in a dynamic process termed angiogenesis.13,14,31,32
Several waves of vessel development and regression occur in proximity to the developing long bones during their transition from mesenchymal primordia (anlagen) to more defined chondral and then ossified structures. There is a limited window of opportunity for vascular invasion of each anlage during its development as endochondral patterning and subsequent ossification ceases after the wave of progress has bypassed that segment of the limb.33,34 The arrangement of vessels prior to the appearance of the primary centers of ossification are present at birth and retained into adulthood.13–15,33,35 Maldevelopment due to aberrant transitioning of these arteries results in corresponding dystrophic post-specification failures of their bony constituencies to form or mineralize.17,20,36
Rapid growth of the lower limb accompanies rapid growth of the embryo from 10 mm crown-rump (CR) length at 5 ½ weeks to 30 mm by the end of the seventh week of development. The primitive Axis Artery (AA) of the developing embryonic limb normally transitions through a series of buddings and regressions to arrive, by the eighth embryonic week, at the more familiar and complex adult arterial pattern of the lower extremity.13–15 The AA branches from the dorsal root of the umbilical artery at the 9 mm CR stage to provide the exclusive blood supply to the lower limb bud,13–15 the tibia posteriorly and the anterior surface of the popliteus muscle as the Deep Popliteal Artery (DPA).13 The femoral artery (FA) grows out from the external iliac artery at the 12 mm CR stage to merge with the AA above the knee by about the 14 mm CR stage.13,14,37 By the 18 mm CR stage, the FA contribution surpasses that of the AA and subsequently serves as the predominant vascular supply below the knee.13–15,37 By the 22 mm CR stage, the continuity of the AA above the knee has been interrupted; it is at this point that only the FA and its derivatives convey blood to the region below the knee, with the exception of the communicating branch of the anterior tibial artery (ATA) (Figure 5).37
Figure 5. Illustration of the habitual transition of arteries supplying the distal limb from the primitive Axial Artery which recedes and is succeeded by the Femoral Artery by the 22 mm CR stage. The segment of the popliteal artery, normally formed by a persistent primitive arterial segment (arrowhead) was atretic in the patient reported;37 the non-primitive segment (arrow) is likely the result of flawed embryonic arterial transitioning. Reproduced with permission.37

As the AA is succeeded by the FA, the DPA is replaced by the Superficial Popliteal Artery (SPA) posterior to the popliteal muscle. After regression of the DPA, the SPA gives rise to the superior and inferior medial and lateral genicular arteries around knee as well as the distal trifurcation of the Anterior Tibial (ATA), Posterior Tibial (PTA), and Peroneal (PA) Arteries, which supply the lower limb.13,14,37 All of the vessels of the trifurcation have been shown to be entirely lacking in CFD (Figure 6).20,23
Figure 6. Illustration of the vascular anatomy of the FfM limb. After the 22 mm CR stage, the femoral artery provides the sole blood supply to the lower limb below the knee. Saphenous and articular branches of the descending genicular artery supply the medial portions of the knee from the medial femoral condyle to the medial tibial plateau. The medial inferior genicular artery remains with the distal portion of the primitive arterial segment.14,32 The descending genicular arteries and the medial inferior genicular artery form independently of the DPA to SPA transition, which selectively affects the lateral superior and lateral inferior genicular arteries as well as the ATA, PTA and PA arteries, which all fail to form (as represented by the dotted lines).23
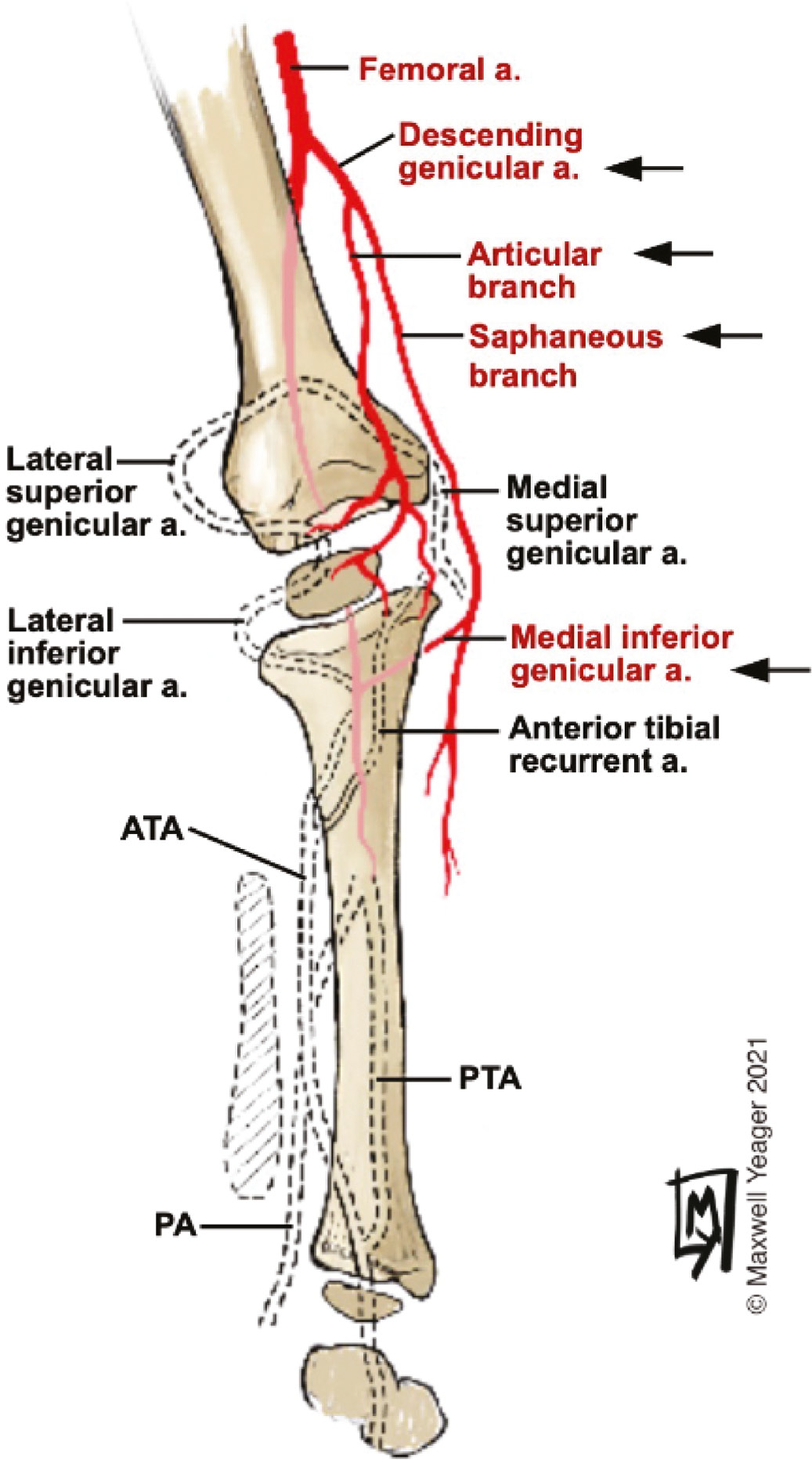
The femoral condyles normally derive their blood supply from the superior medial and lateral genicular branches of the SPA.38,39 The arterial network surrounding the proximal tibia is served principally by branches of the medial and lateral inferior genicular arteries, with contributions from the recurrent branches of the ATA and from the other named genicular arteries, through an anastomosis around the knee joint.38,39 Articular and saphenous branches of the descending genicular artery contribute to the medial side of the anastomosis.15
The distal end of the tibia is a composite bone, formed from a medial (or tibial) part and a lateral (or fibular) part, each with a distinct vascular supply.40,41 The principal vessel serving the medial portion forms by coalescence of a number of descending periosteal arteries (Figure 7). It enters the epiphysis through a notch at the anterior and inferodistal articular surface close to the midline at the interval between the two epiphyses.41 The posterior-lateral parts of the distal tibial epiphysis supplied by circumferential vessels originating as branches of the ATA, PTA, and PA.
Figure 7. A) Antero-posterior, B) lateral and C) coronal illustrations of the epiphysis of the distal tibia from 4A,B, demonstrating the location of the consistent union of periosteal vessels for the interior and medial portions of the tibial plate during development and at skeletal maturity D). This results in the pathological bean-shaped29 bony quarter of the anterior and medial articular surface of the distal tibia. This is evidenced in Figures 3 and 4 as a result of the sole anterior blood supply to the distal tibial epiphysis. Absence of the usual circumferential blood supply in the groove around the distal tibia produced by branches of the ATA, PTA, and PA are missing as evidenced in Figure 6 and the arteriography of the CFD limb in Figure 4.23

Normal Embryonic Development of Long Bones of the Long Bones of the Lower Limb
The lower limb buds first become visible as mesenchymal condensations on the flank of the embryo approximately 4 weeks after conception.11,12,34,42 By the end of the sixth embryonic week, mesenchymal condensations in the core of the limbs are sufficiently molded to resemble their mature bony shapes. Rapid growth of the lower limb accompanies rapid CR growth in length from 10 mm at 5 ½ weeks to 30 mm by the end of the seventh week of development. Specification of the limb segments proceeds in a proximal-to-distal sequence, where the femur is specified before the fibula and tibia and, later, the tarsals, metatarsals, and digits in sequence.33,34 By the beginning of the eighth week, all skeletal elements of the limbs are present as cartilaginous models.33,43
Inter-Relationship of Vascular Transitioning and Growth of the Lower Limb Bones
Specification of the limb segments proceeds in a proximal-to-distal sequence.33,34 The lower limb buds arise as mesenchymal condensations on the flank of the embryo at approximately 4 weeks after conception.11,34 By the end of the sixth embryonic week, mesenchymal condensations are sufficiently molded to resemble their mature bony shapes. By the beginning of the eighth week, all skeletal elements of the limbs are present as cartilaginous anlagen.33,43
Angiogenesis features prominently in normal endochondral development of long bones and, generally, in skeletogenesis, adapting to meet the metabolic demands of the developing bone as it grows.44–47 Blood is supplied to long bones through three primary sources: (a) the diaphyseal nutrient artery, which invades the anlage to establish a primary center of ossification during development and supply the endosteal compartment of the mature meta-diaphysis; (b) the circumferential and epiphyseal arteries, which supply the ends of the long bones to establish secondary ossification centers of the epiphyses as well as the perichondrial rings of Ranvier; and (c) periosteal arteries, which supply both the outer portion of the bone as well as communicate with the endosteal compartment via recently described transcortical vessels.48–51
Ossification of the secondary centers normally occurs perinatally, dividing the epiphyseal anlage into two functionally distinct structures: the articular cartilage of the joint and the physeal growth plate, which continues to drive both longitudinal and latitudinal bone growth postnatally. Prior to its closure at maturity, the physis itself remains avascular; its metabolic demands are met primarily via diffusion of transudate from the terminal branches of the epiphyseal artery and the circumferential vascular network within the ring of Ranvier.49,50,52
Embryonic Arterial Dysgenesis Predicts Multiple Sites of Long Bone Dysmorphism in CFD Limbs
Errors of embryonic arterial development have been associated with a variety of lower limb dysmorphisms.14,20,22,23,33,53,54 Formation of the medial and lateral superior and inferior genicular arteries depend upon normal SPA development (Figure 6). When the DPA to SPA transition has failed, the lateral branches of the genicular artery, ATA, PTA, and PA and their derivatives, as direct branches of the FA, are also lost. However, the more primitive medial inferior genicular as a persisting branch of the AA from the DPA and the superior genicular arteries have medial articular and saphenous branches, which also persist.
The inferior medial genicular and superior genicular arteries continue to supply the medial aspect of the knee independent of dysgenesis of the DPA to SPA transition.32 In contrast, the inferior lateral genicular artery, which requires successful transitioning of the DPA to the SPA, does not form in the CFD limb. Thus, only the lateral superior and inferior genicular arteries, which depend entirely on the normal DPA to SPA transition, fail to form. The pathoanatomy of the arterial tree is evident on angiography of CFD affected limbs which are abnormal in all CSL studied.16,23,30,40,55
As noted earlier, the distal tibial epiphysis is supplied by the circumferential arterial ring within the perichondrial groove of Ranvier, receiving contributions from the usual three vessels of the calf: the ATA, PTA, and PA,48 which are entirely lacking in CFD limbs (Figures 6 and 7).15,24,48,54,56 Failed transition of the DPA to the SPA results in loss of the ATA, PTA, and PA and their contributions to the perichondrium of the distal tibial epiphysis, and therefore failure to initiate a secondary ossification center in the portions of the field that these vessels would have served, with the exception of the anteromedial quadrant.29,40,41 Independent of the DPA to PA transition, this region is served by a solitary vessel formed by the anastomosis of periosteal vessels, which enters the anterior-medial epiphysis through a groove just medial to the midline.41,48 This vessel permits formation of a bean-shaped29 secondary center of ossification in the preserved anteromedial quadrant (as visualized in Figures 3, 4, and 7) radiographically apparent on anteroposterior and lateral views of the distal tibial epiphysis as a wedge- or bean-shaped triangle, tapering toward the midline (Figure 4).8,23,29,41,49
Conclusions
The wide variety of CSL/CFD dysmorphisms deviate from normal limbs, with certain predictable FfM reductions, congruent with specific embryonic vascular dysgeneses as anticipated by the enduring studies of embryonic arterial anatomy by Senior.13,14,32 Epiphyseal reductions of the lateral knee and ankle in affected limbs also conform to specific arterial deficiencies. Embryonic vessels transition via angiogenesis in response to the metabolic demands of the developing long bones they serve. The timing of such transitions is critical in determining the dysmorphologies seen in CSL.13,15,16,20,33 When disturbances occur, the most metabolically active structures later appear dystrophic.18
Lateral knee underdevelopment may respond to guided growth via surgical stapling or epiphysiodesis to prevent medial overgrowth and is indicated to prevent valgus angular deformity of the knee.7 The asymmetric growth of the distal tibial epiphysis, which contributes to the equinovalgus deformity of the foot and ankle in CFD, is not remediable.8 Further examination of embryonic vascular dysgenesis as a common denominator in maldevelopment of the LFC and the lateral parts of the proximal and distal tibial epiphyses and the long bones of the lower limb is thus encouraged.
Acknowledgements
This paper is dedicated to the memory of our dear colleague David S. Packard Jr., PhD, who first originated the term “Post-specification Error of Development.” The authors thank Henry Kronenberg, MD, for his patience in considering our hypotheses, and Amy Slutzky, PhD, for her ongoing bibliographic contributions to this project.
Disclaimer
No funding was received. The authors have no conflicts of interest to disclose pertaining to this manuscript.
References
- O’Rahilly R. Morphological patterns in limb deficiencies and duplications. Am J Anat. 1951;89:135–193.
- Arnold WD. Congenital absence of the fibula. Clin Orthop. 1959;14:20-29.
- Bedoya MA, Chauvin NA, Jaramillo D, et al. Common patterns of congenital lower extremity shortening: diagnosis, classification, and follow-up. Radiographics. 2015;35:1191-1207.
- Achterman C, Kalamchi A. Congenital deficiency of the fibula. J Bone Joint Surg Br. 1979;61-B:133-137.
- Birch JG, Lincoln TL, Mack PW, et al. Congenital fibular deficiency: a review of thirty years’ experience at one institution and a proposed classification system based on clinical deformity. J Bone Joint Surg Am. 2011;93:1144-1151.
- Rodriguez-Ramirez A, Thacker MM, Becerra LC, et al. Limb length discrepancy and congenital limb anomalies in fibular hemimelia. J Pediatr Orthop B. 2010;19:436-440.
- Boakes JL, Stevens PM, Moseley RF. Treatment of genu valgus deformity in congenital absence of the fibula. J Pediatr Orthop. 1991;11:721-724.
- Choi IH, Lipton GE, Mackenzie W, et al. Wedge-shaped distal tibial epiphysis in the pathogenesis of equinovalgus deformity of the foot and ankle in tibial lengthening for fibular hemimelia. J Pediatr Orthop. 2000;20:428-436.
- Sanpera I, Jr., Sparks LT. Proximal femoral focal deficiency: does a radiologic classification exist? J Pediatr Orthop. 1994;14:34-38.
- Freund E. Congenital defects of femur, fibula, tibia. Arch Surg. 1936;33:349-391.
- Patten BM. Human Embryology. Philadelphia, PA: McGraw-Hill; 1953.
- Sadler TW. Langman’s Medical Embryology. Philadelphia: Wolters-Kluwer; 2019.
- Senior HD. The development of the arteries of the human lower extremity. Am J Anat. 1919;25:54-95.
- Senior HD. An interpretation of the recorded arterial anomalies of the human leg and foot. J Anat. 1919;53:130-171.
- Senior HD. Development of the arteries. In: Clemente C, ed. Baltimore, M.D.: Williams and wilkins; 1985.
- Hootnick DR, Vargesson N. The syndrome of proximal femur, fibula, and midline metatarsal long bone deficiencies. Birth Defects Res. 2018;110:1188-1193.
- Packard DS, Jr., Levinsohn EM, Hootnick DR. Most human lower limb malformations appear to result from postspecification insults. Prog Clin Biol Res. 1993;383A:417-426.
- Stockard CR. Developmental Rate and Structural Expression: an experimental study of twins, ‘double monsters’ and single deformities, and the interaction among embryonic organs during their origin and development. Am J Anat. 1921;28:115-277.
- Hamanishi C. Congenital short femur. Clinical, genetic and epidemiological comparison of the naturally occurring condition with that caused by thalidomide. J Bone Joint Surg Br. 1980;62:307-320.
- Hootnick DR. Congenital fibular dystrophisms conform to embryonic arterial dysgenesis. Anat Rec (Hoboken). 2020;303:2792-2800.
- Hootnick DR. Missing midline metatarsals conform to plantar arterial arch dysgenesis. J Am Podiatr Med Assoc. 2020;110.
- Chomiak J, Horak M, Masek M, et al. Computed tomographic angiography in proximal femoral focal deficiency. J Bone Joint Surg Am. 2009;91:1954-1964.
- Hootnick DR, Levinsohn EM, Randall PA, Packard DS, Jr. Vascular dysgenesis associated with skeletal dysplasia of the lower limb. J Bone Joint Surg Am. 1980;62:1123-1129.
- Hootnick DR, Levinsohn EM, Packard DS, Jr. Midline metatarsal dysplasia associated with absent fibula. Clin Orthop Relat Res. 1980:203-206.
- Thompson TC, Straub LR, Arnold WD. Congenital absence of the fibula. J Bone Joint Surg Am. 1957;39-A:1229-1237.
- Bohne WH, Root L. Hypoplasia of the fibula. Clin Orthop Relat Res. 1977:107-112.
- Westin GW, Sakai DN, Wood WL. Congenital longitudinal deficiency of the fibula: follow-up of treatment by Syme amputation. J Bone Joint Surg Am. 1976;58:492-496.
- Davidson WH, Bohne WH. The Syme amputation in children. J Bone Joint Surg Am. 1975;57:905-909.
- Dieulafe R, Cahuzac M. Un cas d’absence partielle congenitale due perone avec dissection. Bord Chir. 1935;6:196-210.
- Hootnick DR, Levinsohn EM. Embryology of the lower limb demonstrates that congenital absent fibula is a radiologic misnomer. Anat Rec (Hoboken). 2022;305:8-17.
- Ambler CA, Nowicki JL, Burke AC, et al. Assembly of trunk and limb blood vessels involves extensive migration and vasculogenesis of somite-derived angioblasts. Dev Biol. 2001;234:352-364.
- Senior HD. Abnormal branching of the human popliteal artery. Am J Anat. 1929;44:111-120.
- Vargesson N, Hootnick DR. Arterial dysgenesis and limb defects: clinical and experimental examples. Reprod Toxicol. 2017;70:21-29.
- Tickle C. How the embryo makes a limb: determination, polarity and identity. J Anat. 2015;227:418-430.
- Taylor GI, Palmer JH. The vascular territories (angiosomes) of the body: experimental study and clinical applications. Br J Plast Surg. 1987;40:113-141.
- Dirckx N, Van Hul M, Maes C. Osteoblast recruitment to sites of bone formation in skeletal development, homeostasis, and regeneration. Birth Defects Res C Embryo Today. 2013;99:170-191.
- Neville RF, Jr., Franco CD, Anderson RJ, et al. Popliteal artery agenesis: a new anatomic variant. J Vasc Surg. 1990;12:573-576.
- Crock HV, Crock MC. The lower end of the femur. In: Crock HV, ed. Edinburgh, U.K.: E&S Livingstone; 1967:29-46.
- Crock HV, Crock MC. The upper end of the tibia. In: Crock HV, ed. Edinburgh, U.K.: E&S Livingstone; 1967:55-63.
- Trueta J. The Development of the Epiphyses. London, U.K.: William Heineman Medical Books LTD; 1968:84-89.
- Morgan JD. Blood supply of growing rabbit’s tibia. J Bone Joint Surg Br. 1959;41-B:185-203.
- O’Rahilly R, Muller F. Human Embryology & teratology. New York, NY: Wiley-Liss; 1992.
- O’Rahilly R, Gardner E. The timing and sequence of events in the development of the limbs in the human embryo. Anat Embryol (Berl). 1975;148:1-23.
- Maes C. Role and regulation of vascularization processes in endochondral bones. Calcif Tissue Int. 2013;92:307-323.
- Maes C. Signaling pathways effecting crosstalk between cartilage and adjacent tissues: seminars in cell and developmental biology: the biology and pathology of cartilage. Semin Cell Dev Biol. 2017;62:16-33.
- Kusumbe AP, Ramasamy SK, Adams RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014;507:323-328.
- Kronenberg HM. Developmental regulation of the growth plate. Nature. 2003;423:332-336.
- Trueta J. The Origin of the Circulatory System. London, U.K.: William Heineman Medical Books LTD; 1968:69-83.
- Trueta J, Morgan JD. The vascular contribution to osteogenesis. I. Studies by the injection method. J Bone Joint Surg Br. 1960;42-B:97-109.
- Rang M. The Growth Plate and its Disorders. Edinburgh, U.K.: E&S Livinstone; 1969.
- Gruneboom A, Hawwari I, Weidner D, et al. A network of trans-cortical capillaries as mainstay for blood circulation in long bones. Nat Metab. 2019;1:236-250.
- Trueta J. The role of the vessels in osteogenesis. J Bone Joint Surg. 1963;45-B:402-418.
- Senior HD. An interpretation of the recorded arterial anomalies of the human pelvis and thigh. Am J Anat. 1925;36:1-46.
- Levinsohn EM, Hootnick DR, Packard DS, Jr. Consistent arterial abnormalities associated with a variety of congenital malformations of the human lower limb. Invest Radiol. 1991;26:364-373.
- Hootnick DR, Packard DS, Jr., Levinsohn EM, et al. The anatomy of a congenitally short limb with clubfoot and ectrodactyly. Teratology. 1984;29:155-164.
- Crock HV, Crock MC. The lower end of the tibia. In: Crock HV, ed. Edinburgh, U.K.: E&S Livingstone; 1967:72-75.
