Original Research
Retrospective Review of 181 Patients with Pathologic Vertebral Compression Fractures
1Baylor College of Medicine, Houston, TX; 2Texas Children’s Hospital, Houston, TX
Correspondence: Tristen N. Taylor, BS, 1 Hermann Museum Circle Dr. 4037, Houston, TX 77004. E-mail: [email protected]
Received: March 28, 2023; Accepted: May 10, 2023; Published: August 1, 2023
Volume 5, Number 3, August 2023
Abstract
Background: Compression fractures are often associated with lower energy trauma and may occur in the setting of abnormal bone health associated with genetic disorders and endocrine disorders, neoplastic disease, infection, and inflammatory disorders. There is no significant series in the literature describing the prevalence or etiology of pathologic pediatric compression fractures.
Methods: IRB-approved retrospective study was performed at a tertiary children’s hospital from 2012-2022. Patients <18 years old diagnosed with atraumatic vertebral compression fractures were included and reviewed for demographics, underlying diagnosis/comorbidity, presentation, mobility, deformities, imaging data, treatments, and outcomes.
Results: 181 patients (54% Male) were included with mean age 14.17 years and follow-up of 20 months. A compression fracture was the presenting symptom of an underlying diagnosis in 32% of patients, and 21% of patients received an MRI to distinguish between metastatic disease and benign fractures. Primary osteoporosis was the cause in 15% and secondary osteoporosis was in 65% of patients; most commonly due to immunosuppressants (46%) and acute lymphoblastic leukemia (ALL) (10%). Primary lesions were the etiology in 20% of patients. There was a median of 3 fractures per patient, mostly of the midthoracic (82%) and thoracolumbar spine (51%). Radiographs revealed wedge fractures in 82%, biconcave fractures in 22%, and vertebra plana in 12%. Patients were managed with observation (46%), bracing (31%), bisphosphonates (31%), and fusions (4%). By last follow-up, there was a higher proportion of patients who developed scoliosis or became wheelchair-bound. Overall, there was a 16% mortality rate which was mostly associated with cancer.
Conclusion: Overall, 32% of patients presented with a compression fracture as the presenting symptom of an underlying disease. Pathologic vertebral compression fractures in children frequently occurred due to immunosuppressants, ALL, and metastatic disease. The fractures were most often wedge morphology in the thoracolumbar and mid-thoracic regions of the spine. MRIs may be useful for distinguishing between benign fractures and malignancy.
Levels of Evidence: Level IV
Key Concepts
- Pathologic Vertebral Compression Fractures occur commonly in children with primary or secondary osteoporosis or due to primary site lesions, such as metastases.
- Patients often have multiple fractures about the thoracolumbar and mid-thoracic regions of the spine.
- Wedge and biconcave fractures were the most common fracture type at presentation and indicated an underlying disease not previously known in one-third of patients.
- MRI is useful for distinguishing between benign fractures and malignancy as the etiology of the fracture.
Introduction
Pediatric spine fractures are rare in occurrence, accounting for 1-2% of all pediatric fractures.1 Of all pediatric spine fractures, compression fractures are the most prevalent.2 While the majority of compression fractures are traumatic, they may also occur in the setting of abnormal bone health associated with genetic and endocrine conditions, neoplastic disease, infection, or inflammatory disorders.1,2
There has been greater emphasis on the recognition and management of osteoporosis in children over the last few decades.3,4 Primary osteoporosis occurs due to an underlying genetic defect, most commonly Osteogenesis Imperfecta, but may also be idiopathic.5 Secondary osteoporosis occurs due to a systemic underlying condition, like hypercortisolism, acute lymphocytic leukemia (ALL), or by immobility.3 Secondary osteoporosis also frequently occurs from medications, like glucocorticoids or chemotherapy, which suppresses bone turnover.6 Children with osteoporosis often present with recurrent fractures and back pain, and the incidental finding of a compression fracture may lead to a diagnosis of osteoporosis.3 Pathologic compression fractures in children may also occur by structural compromise through a primary lesion of the vertebral body. Etiologies such as metastatic disease, Chronic Recurrent Multifocal Osteomyelitis (CRMO), and Langerhans Cell Histiocytosis (LCH) frequently present with vertebral lesions and may even result in vertebra plana.7–10
Because pediatric vertebral compression fractures may result in significant height loss, deformities, chronic pain, and may indicate an underlying pathology, it is important to recognize and appropriately manage these patients.11–13 Despite the multifaceted etiology of pathologic pediatric compression fractures and its effects, to our knowledge, there has been no significant series in the literature to describe these patients. Therefore, we aimed to review the etiologies, fracture characteristics, and outcomes of children diagnosed with pathologic vertebral compression fractures.
Methods
After IRB approval, we conducted a retrospective review of patients diagnosed with a vertebral compression fracture of the spine by ICD-10 code (M48.5-). Patients were included if they were diagnosed with a vertebral compression fracture before age 18 with at least 6 months of follow-up. Patients were excluded if their fracture occurred after age 18, did not meet minimum follow-up, or if the compression fractures were traumatic etiology.
Patient charts were reviewed for demographic data (age, follow-up time, gender, BMI), underlying etiology, pain, neurologic symptoms, mobility (community walker, home-walker, or wheelchair-bound), and the number of patients who initially presented with a compression fracture and were subsequently diagnosed with an underlying etiology. Outcomes and treatment data such as management with observation, bracing, fusion, vertebroplasty, NSAIDs, bisphosphonates, opiate use, date of death and time from fracture diagnosis to death were also obtained.
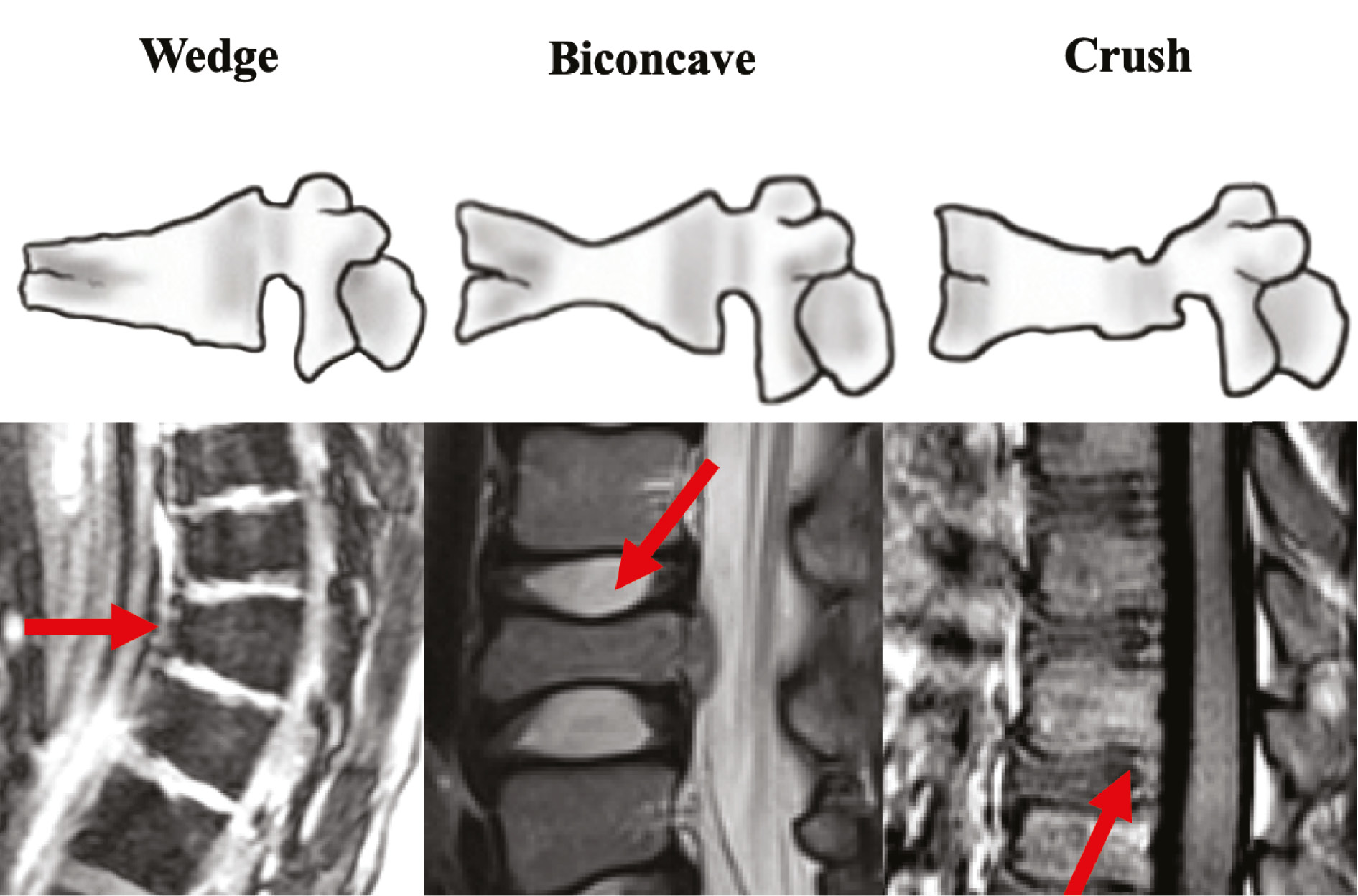
Radiographic and MRI reports were reviewed for presence of scoliosis or kyphosis, number of fractures, and location. Fractures were characterized as wedge, pincer/split, burst, or vertebra plana (Figure 1).14 The indication to distinguish between malignancy and benign compression fracture were also recorded from the MRI report or the ordering physician’s plan. Imaging at last follow-up was evaluated for scoliosis and kyphosis progression, vertebral height change, and new compression fractures.
Figure 1. Vertebral compression fracture types. Fracture shapes are classified based on whether the anterior (wedge), middle (biconcave), or posterior (crush) columns was primarily involved.
Statistical Analysis
Descriptive data was reported with count and percentage, mean and standard deviation for parametric data, or median and range for non-parametric data. The McNemar test was used to compare the proportion of patients with pain, neurologic symptoms, mobility, scoliosis, and kyphosis at last follow-up vs. at presentation; the odds ratio, 95% confidence interval, and P-value were reported. Underlying etiology in deceased patients was compared with the Chi-Square test, and relative risk (RR) was reported with 95% confidence interval. A P-value of 0.05 was designated as significant.
Results
Demographics
Of the 502 patients collected, 321 patients were excluded due to age, low follow-up, or traumatic fracture. Therefore, this study included 181 patients, 97 males and 84 females, with an average age (SD) of 14 ± 7.37 years and median follow-up of 20 (median: 6-41) months. The mean BMI was 22.49 ± 7.37 kg/m2.
Etiology
In 58 (32%) patients, a compression fracture was the presenting symptom or radiographic finding of an underlying diagnosis that was not previously known. Overall, 15% of patients had primary osteoporosis (Table 1), and 118 (65%) of patients had secondary osteoporosis, most commonly due to immunosuppressants (46%) and ALL (10%). Metastases were present in 19 (10%) of fractures and were from neuroblastomas (5 patients, 3%), Ewing Sarcoma (3, 2%), lymphoma (2, 1%), osteosarcoma (2, 1%), rhabdomyosarcoma (2, 1%), hepatoblastoma (1, 0.5%), leiomyosarcoma (1, 0.5%), pineal germinoma (1, 0.5%), renal medullary carcinoma (1, 0.5%), and spindle cell carcinoma of the mandible (1, 0.5%). Other causes of compression fractures included LCH, CRMO, an aneurysmal bone cyst, and fibrous dysplasia. There were no compression fractures associated with infection.
Table 1. Vertebral Compression Fracture Etiologies
| Etiology | Count | %* |
|---|---|---|
| Primary Osteoporosis | 27 | 15% |
| Idiopathic | 12 | 7% |
| Osteogenesis Imperfecta | 12 | 7% |
| Other connective tissue disorder | 3 | 2% |
| Secondary Osteoporosis | 118 | 65% |
| Acute Lymphoblastic Leukemia | 19 | 10% |
| Received Immunosuppressants | 83 | 46% |
| Malabsorption | 12 | 7% |
| Immobility | 4 | 2% |
| Primary lesion | 36 | 20% |
| Neoplasm | 33 | 18% |
| Metastases | 19 | 10% |
| Langerhans Cell Histiocytosis | 12 | 7% |
| Aneurysmal Bone Cyst | 1 | 1% |
| Fibrous dysplasia | 1 | 1% |
| CRMO | 3 | 2% |
CRMO = Chronic recurrent multifocal osteomyelitis; *n = 181.
Fracture Characteristics
There were 579 fractures, with a median of 3 (range: 1-12) fractures per patient (Figure 2). 148 (82%) patients had thoracic, 92 (51%) had lumbar, and 8 (4%) had cervical fractures. L1 (12%), T12 (11%), T8 (8%), L2 (8%), and T6 (7%) were the most common sites of compression fracture. Radiographs (Table 2) revealed wedge fractures in 82% of patients, biconcave fractures in 22%, and vertebra plana in 12%. Eleven (8%) patients presented with radiographic evidence of scoliosis, and 22 (14%) patients presented with kyphosis. MRI revealed concurrent lesions or fracture involvement of the epidural space in 20%, retropulsion in 18%, and disc herniation in 6%. MRI was also ordered in 21% of patients to specifically distinguish between metastatic disease and benign compression fracture. At last follow-up, imaging revealed an additional compression fracture in 15% of patients and vertebral body height improvement in 29%. Patients with secondary osteoporosis (RR: 2.6; 95% CI: 1.9-3.5; P<0.001), metastases (RR: 5.3; CI: 2.2-15.4; P<0.001), and LCH (RR: 3; CI: 1.2-8.7; P=0.04) were less likely to vertebral height improvement.
Figure 2. Total count of pathologic compression fractures per spinal level in 181 patients.

Table 2. Imaging Findings
| Finding | Count | % |
|---|---|---|
| Radiograph | 158 | 87%* |
| Wedge | 129 | 82% |
| Biconcave | 35 | 22% |
| Crush | 18 | 11% |
| Vertebra Plana | 19 | 12% |
| Osteoporosis/Osteopenia | 67 | 42% |
| Osteolysis | 21 | 13% |
| Sclerosis | 20 | 13% |
| Kyphosis | 22 | 14% |
| Scoliosis | 11 | 8% |
| MRI | 76 | 42%* |
| Wedge | 60 | 79% |
| Biconcave | 43 | 57% |
| Crush | 14 | 18% |
| Lesions involving Epidural Space | 15 | 20% |
| Retropulsion | 14 | 18% |
| Disc Herniation | 6 | 8% |
MRI = Magnetic Resonance Imaging; LCH = Langerhans Histiocytosis; *n = 181; Subcategories of Radiograph n=158; MRI, n=76.
Outcomes
Patients were managed with observation (46%), bracing (31%), or bisphosphonates (31%). Overall, 8 (4%) of patients required surgical intervention: 5 required posterior spinal fusions to stabilize their hyperkyphosis or scoliosis, and 3 required anterior cervical discectomy and interbody fusion (ACDF) due to primary lesions causing vertebra plana, hyperkyphosis, and spinal cord compression. Patients were treated with NSAIDs (56%), and 30% received opioids. At diagnosis, 58% patients reported back pain, and 14% of patients had neurologic symptoms: 9% had lower extremity weakness, 6% had loss of sensation, and 3% had incontinence. Between primary osteoporosis, secondary osteoporosis, and primary lesions, no etiology had a relatively greater risk of having pain, scoliosis, kyphosis, or becoming wheelchair-bound. Compared to the proportion of patients at presentation, at last follow-up there were significantly less patients with pain (38% vs. 19%; OR 8.9; CI 4.4-20; P<.001) and kyphosis (12% vs. 8%; OR 3.4; CI 1.4-9.4; P=.003). However, more patients developed scoliosis (8% vs. 12%; OR 0.375; CI 0.12-.99; P=.03) or were limited to a wheelchair (7% vs. 15%; OR 0.16; CI 0.03-0.54; P<.001). Overall, 29 (16%) patients were deceased at the time of data collection after a mean 31.4 months (range: 2.6-108 months). Of the deceased patients, 19 had cancer (RR: 1.9; CI: 1.1-3.4, P=.034), 2 had Duchenne Muscular Dystrophy (DMD), 2 had cystic fibrosis, and 6 had various genetic disorders.
Discussion
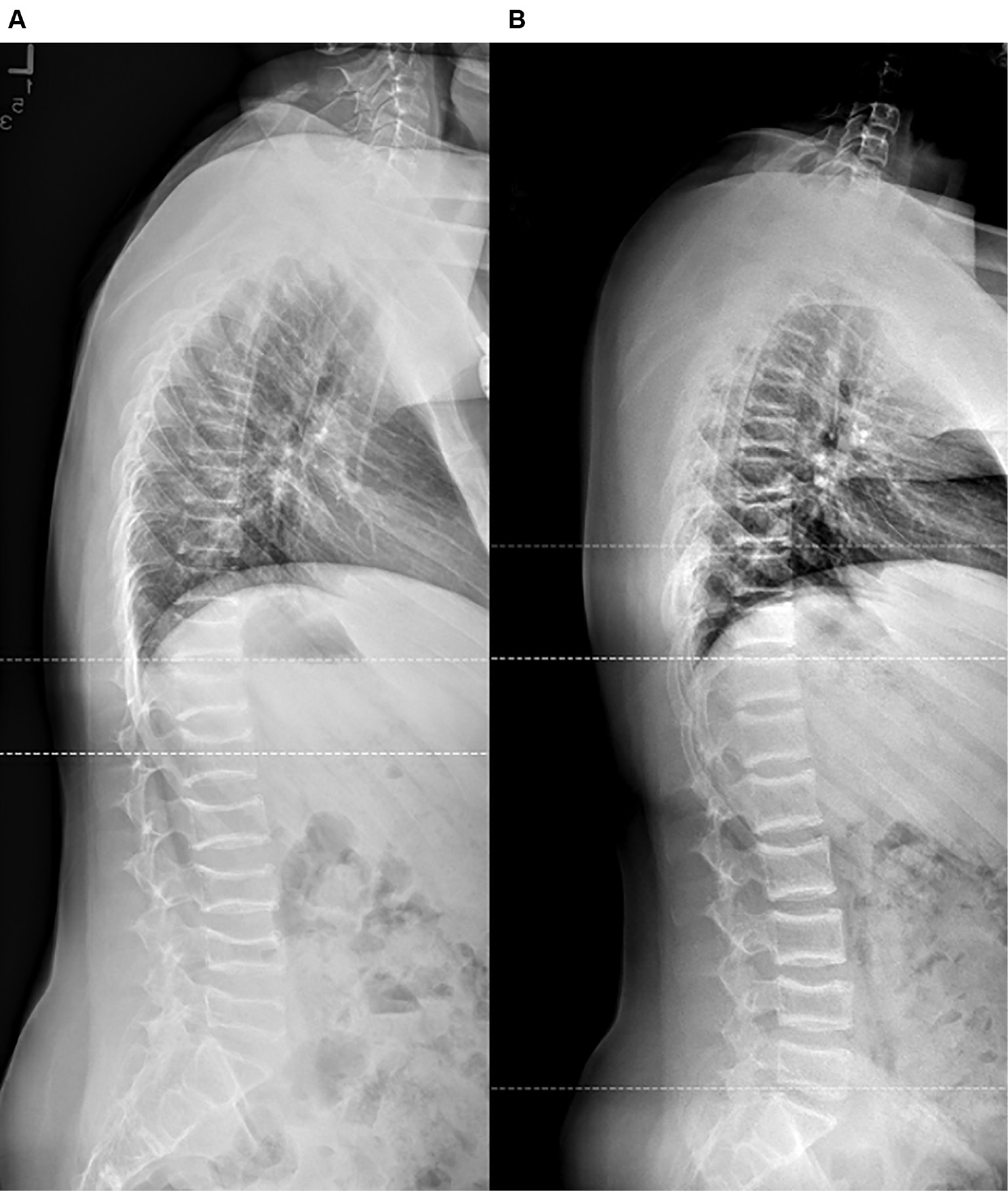
This retrospective study described the clinical and fracture characteristics, etiologies, and outcomes of 181 patients with pathologic vertebral compression fractures (Figure 3). Secondary osteoporosis was the most common etiology of compression fracture. Fractures were diagnosed through a combination of radiographs and MRI and were mostly wedge or biconcave fractures involving the thoracolumbar junction. By last follow-up, patients had better pain control but worsened mobility and scoliosis. Patients were predominantly managed medically, and the mortality rate of our cohort was 16% (29 patients), which was highly associated with cancer (P=.034).
Figure 3. A) Lateral spinal radiograph of a 13-year-old male patient with marked osteopenia due to chemotherapy, loss of vertebral body heights due to wedge and biconcave compression fractures involving the near-entire thoracic and lumbar spine, and significant thoracic kyphosis. B) The same patient 4 years later after zoledronic acid and bracing treatments. Endplate sclerosis and multiple biconcave compression deformities still exist, though vertebral height and thoracic kyphosis have significantly improved.
Immunosuppressants or ALL were the leading cause of secondary osteoporotic fractures in a combined 65% of our cohort. Prior to diagnosis, 46% of our cohort received immunosuppressants, whereas only 7% and 2% were associated with malabsorption and immobility, respectively. While prolonged or high dose immunosuppressants are the most common etiology of osteoporosis in patients with cancer, ALL is also a unique independent cause of osteoporotic fractures in children.15–18 Osteoclast-activating cytokines released from leukemic cells cause cortical thinning, endosteal resorption, and ultimately osteoporosis.3,18,19 Alos et al. found a fracture prevalence of 16% (n=155) among newly diagnosed ALL patients,19 and 10% of patients in our cohort presented with a compression fracture secondary to ALL. Because of the association between vertebral compression fractures and chronic immunosuppression or ALL, monitoring of bone health and vertebral fractures should be part of a standard follow-up evaluation for these at-risk patients.
Primary lesions cause compression fractures under normal physiologic stress by destabilizing a portion of the vertebral body20,21 and were associated with 18% of patients. Metastatic disease was shown to be associated with compression fractures, as Sinha et al. found pathologic fractures in 4 of 20 (20%) children with metastases.13 Spinal metastases were present in 10% of children with compression fractures and has previously been reported to be associated with 39% of all bony metastases by Baker et al.22 Primary lesions causing compression fractures from rare diseases CRMO and LCH were also present in 9% of our cohort. Though very different in pathophysiology and clinical presentation, these diseases may cause multifocal osteolytic lesions of vertebral bodies and vertebra plana.7,23 Because metastatic lesions, CRMO, and LCH risk being misdiagnosed as benign osteoporotic fractures,8,10 close serial monitoring via MRI or bone biopsy should be performed to confirm their diagnosis. Appropriate referral to rheumatologists for CRMO and oncologists for metastatic disease or LCH, is crucial to prevent further spinal deterioration.10
As the diagnosis of osteoporosis in children can be made by the presence of one or more vertebral compression fractures in the absence of local disease or trauma,4 it is important they are efficiently characterized. Similar to Halton et al., our cohort’s fractures were distributed about the thoraco-lumbar junction and mid-thoracic region.18 Compressive forces are maximized around these areas because the mid-thoracic region is where the thoracic curve is the greatest in the sagittal plane, and the thoraco-lumbar junction is where the spine transitions from being fixed to ribs.24 A lateral spine radiograph is the first line imaging study for evaluating for compression fractures. Wedge and biconcave fractures were the most common presentation of fractures, whereas adults presented most frequently with wedge and crush fractures.24 MRIs were also useful for discriminating between metastatic lesions and benign compression fractures, as this was the indication for ordering an MRI for 21% of patients in our cohort. MRIs were able to show local consequences of the fracture through retropulsion, herniation, or epidural involvement. Lateral spine imaging and MRIs proved to be useful imaging modalities for monitoring and detect compression fractures in children at risk of poor bone health.
Compression fractures of the vertebrae may initially be painless as only 58% of our cohort initially reported back pain, which was similar to Halton et al. (55%).18 Conversely, 32% of patients presented for chronic back pain due to a previously unknown pathologic compression fracture and were subsequently diagnosed with an underlying etiology. By the last follow-up, pain was significantly decreased as the fractures healed and the underlying illness was treated. However, there were also more patients wheelchair-bound or who developed scoliosis. It is difficult to distinguish between the pathologic fracture and underlying disease as the primary reason for the loss of mobility in patients, though multiple contiguous compression fractures are associated with scoliosis development by destabilizing the structural integrity of the vertebral body, disposing the spine to a coronal or sagittal curvature. Because pathologic fractures are often asymptomatic at presentation, may indicate an underlying disease, and can result in worsening mobility and deformity,18,25 protocols that emphasize a lower threshold for obtaining spinal imaging should be considered to prevent worse outcomes and diagnose underlying disease.26
Many of our patients were managed conservatively with observation or bracing, though 4% required fusions for sequelae of their vertebral fracture, namely hyperkyphosis secondary to vertebra plana. Surgical interventions for pediatric pathologic vertebral fractures have not been reported on, though Lastikka et al. reported a 33% complication rate and 11% revision risk in children receiving cervical fusions for skeletal dysplasia or trauma,27 whereas Erfani et al. found favorable functional outcomes in pediatric patients receiving spinal fusion for traumatic vertebral fractures.28 Furthermore, although AAOS strongly recommended against vertebroplasty in 2017 for adults with osteoporotic compression fractures,29 vertebroplasty has been shown to at least be a viable palliative treatment option in children.30–32 Although no outcome or comparison data is available in children with pathologic vertebral compression fractures, our practice strongly recommends bracing children at risk of progressive severe scoliosis or kyphosis, frequent follow-up, and appropriate operations in select patients who fail conservative management.
There are insufficient studies on the effect of long-term bisphosphonate use in pediatrics, though short-term bisphosphonates appear to improve bone density and pain management in children with osteoporotic fractures.33,34 Multicenter randomized control trials of bisphosphonate use with chemotherapy for osteosarcomas may lead to a standard protocol for these patients (French OS2006- NTC00470223). In our study, 16% of patients were deceased after a mean 31.4 months, primarily due to metastatic disease or leukemia. While compression fractures are associated with a mortality risk in the geriatric population,35 the influence of pathologic vertebral fractures on mortality in children is currently unknown. Research into pediatric pathologic compression fracture prevention, treatment, interventions, and influence on mortality should be pursued to gauge risk and potentially improve outcomes.
This study is not without limitations. Our series did not report on patient-reported outcomes, so we are limited to outcomes from the last follow-up visit. Like all retrospective series, our study is prone to measurement bias which was avoided through multiple chart reviewers. Despite these limitations, our study identifies the etiologies, imaging characteristics, and outcomes of pathologic vertebral compression fractures. Greater emphasis on management of vertebral fractures should be taken to prevent deformity, evaluate for neoplasia, and limit mortality.
Conclusion
Pathologic vertebral compression fractures in children frequently occur due to immunosuppressants, ALL, metastatic disease, CRMO, and LCH. Wedge and biconcave fractures in the thoracolumbar and mid-thoracic regions of the spine were most common. As 32% of patients presented with a compression fracture as the presenting symptom or radiographic finding of an underlying disease not previously known, MRIs may be useful for distinguishing between benign fractures and malignancy. Future research on the mortality risk, prevention, treatment, and surgical interventions for pathologic fractures should be pursued to aid these patients.
Additional Links
- POSNA Study Guide: Thoracolumbar Spine Fractures
- POSNA Study Guide: Osteogenesis Imperfecta
Disclaimer
No funding was received. The authors report no conflicts of interest related to this manuscript.
References
- Basu S. Spinal injuries in children. Front Neurol. 2012;3:96.
- Daniels AH, Sobel AD, Eberson CP. Pediatric thoracolumbar spine trauma. J Am Acad Orthop Surg. 2013;21(12):707-716.
- Ciancia S, van Rijn RR, Högler W, et al. Osteoporosis in children and adolescents: when to suspect and how to diagnose it. Eur J Pediatr. 2022;181(7):2549-2561.
- Bishop N, Arundel P, Clark E, et al. Fracture prediction and the definition of osteoporosis in children and adolescents: the ISCD 2013 Pediatric Official Positions. J Clin Densitom. 2014;17(2):275-280.
- Forlino A, Cabral WA, Barnes AM, et al. New perspectives on osteogenesis imperfecta. Nat Rev Endocrinol. 2011;7(9):540-557.
- Saraff V, Högler W. ENDOCRINOLOGY AND ADOLESCENCE: Osteoporosis in children: diagnosis and management. Eur J Endocrinol. 2015;173(6):R185-R197.
- Nakamura N, Inaba Y, Aota Y, et al. Characteristic reconstitution of the spinal langerhans cell histiocytosis in young children. J Pediatr Orthop. 2019;39(4):e308-e311.
- Kim SG, Ju CI, Wang HS, et al. Malignant metastasis misdiagnosed as osteoporotic compression fracture: a case report. Korean J Neurotrauma. 2018;14(1):39-42.
- Hospach T, Langendoerfer M, von Kalle T, et al. Spinal involvement in chronic recurrent multifocal osteomyelitis (CRMO) in childhood and effect of pamidronate. Eur J Pediatr. 2010;169(9):1105-1111.
- Taylor TN, Bridges CS, Ezeokoli EU, et al. Retrospective review of 80 patients with chronic recurrent multifocal osteomyelitis evaluated by pediatric orthopaedic surgeons: original research. J Pediatr Orthop Soc N Am. 2023;5(1). https://doi.org/10.55275/JPOSNA-2023-575.
- Saul D, Dresing K. Epidemiology of vertebral fractures in pediatric and adolescent patients. Pediatr Rep. 2018;10(1):7232.
- Sabo A, Hatgis J, Granville M, et al. Multilevel contiguous osteoporotic lumbar compression fractures: the relationship of scoliosis to the development of cascading fractures. Cureus. 2017;9(12):e1962.
- Sinha AK, Seki JT, Moreau G, et al. The management of spinal metastasis in children. Can J Surg. 1997;40(3):218-226.
- Genant HK, Wu CY, van Kuijk C, et al. Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res. 1993;8(9):1137-1148.
- Tantawy AAG, El Bostany EA, Matter RM, et al. Bone mass and biochemical markers of bone turnover in children and adolescents with chronic immune thrombocytopenia: relation to corticosteroid therapy and vitamin D receptor gene polymorphisms. Platelets. 2013;24(4):282-287.
- Dilber C, Dagdemir A, Albayrak D, et al. Reduced bone mineral density in childhood chronic idiopathic thrombocytopenic purpura treated with high-dose methylprednisolone. Bone. 2004;35(1):306-311.
- Huber AM, Gaboury I, Cabral DA. et al. Prevalent vertebral fractures among children initiating glucocorticoid therapy for the treatment of rheumatic disorders. Arthritis Care Res (Hoboken). 2010;62(4):516-526.
- Halton J, Gaboury I, Grant R, et al. Advanced vertebral fracture among newly diagnosed children with acute lymphoblastic leukemia: results of the canadian STeroid-associated osteoporosis in the pediatric population (STOPP) research program. J Bone Miner Res. 2009;24(7):1326-1334.
- Alos N, Grant R, Ramsay T, et al. High incidence of vertebral fractures in children with acute lymphoblastic leukemia 12 months after the initiation of therapy. J Clin Oncol. 2012;30(22):2760-2767.
- Krishnaney AA, Steinmetz MP, Benzel EC. Biomechanics of metastatic spine cancer. Neurosurg Clin N Am. 2004;15(4):375-380.
- Dimar JR, Voor MJ, Zhang YM, et al. A human cadaver model for determination of pathologic fracture threshold resulting from tumorous destruction of the vertebral body. Spine (Phila Pa 1976). 1998;23(11):1209-1214.
- Baker LL, Goodman SB, Perkash I, et al. Benign versus pathologic compression fractures of vertebral bodies: assessment with conventional spin-echo, chemical-shift, and STIR MR imaging. Radiology. 1990;174(2):495-502.
- Khanna G, Sato TSP, Ferguson P. Imaging of chronic recurrent multifocal osteomyelitis. Radiographics. 2009;29(4):1159-1177.
- Ismail AA, Cooper C, Felsenberg D, et al. Number and type of vertebral deformities: epidemiological characteristics and relation to back pain and height loss. Osteoporos Int. 1999;9(3):206-213.
- Cummings EA, Ma J, Fernandez CV, et al. Incident vertebral fractures in children with leukemia during the four years following diagnosis. J Clin Endocrinol Metab. 2015;100(9):3408-3417.
- Ward LM. Part I: Which child with a chronic disease needs bone health monitoring? Curr Osteoporos Rep. 2021;19(3):278-288.
- Lastikka M, Aarnio J, Helenius I. Instrumented cervical spinal fusions in children: indications and outcomes. J Child Orthop. 2017;11(6):419-427.
- Erfani MA, Pourabbas B, Nouraie H, et al. Results of fusion and instrumentation of thoracic and lumbar vertebral fractures in children: a prospective ten-year study. Musculoskelet Surg. 2014;98(2):107-114.
- Genev IK, Tobin MK, Zaidi SP, et al. Spinal compression fracture management. Global Spine J. 2017;7(1):71-82.
- Missiuna PC, Sarraj M, Bosakhar B, et al. Vertebroplasty for treatment of steroid-induced osteoporotic vertebral compression fractures in an adolescent with duchenne muscular dystrophy. HSS Journal®. 2021;17(2):223-226.
- Carbó E, Riquelme Ó, García A, et al. Vertebroplasty in a 10-year-old boy with Gorham-Stout syndrome. Eur Spine J. 2015;24(Suppl 4):S590-S593.
- Leite LC, Rassekh S, D’Ortenzio R, et al. Vertebroplasty as a Palliative Treatment Option for Intractable Pain in Pediatric Patients with Spinal Tumors. Preprints; 2021.
- Eghbali-Fatourechi G. Bisphosphonate therapy in pediatric patients. J Diabetes Metab Disord. 2014;13:109.
- Boyce AM, Tosi LL, Paul SM. Bisphosphonate treatment for children with disabling conditions. PM R. 2014;6(5):427-436.
- Lau E, Ong K, Kurtz S, et al. Mortality following the diagnosis of a vertebral compression fracture in the Medicare population. J Bone Joint Surg Am. 2008;90(7):1479-1486.
