Quality, Safety, and Value (QSVI)
A Quality Improvement Initiative to Reduce Opioid Prescriptions Following Surgical Treatment of Supracondylar Humerus Fractures in Children
1University of California, San Diego, San Diego, CA; 2Department of Orthopedic Surgery, Rady Children’s Hospital, San Diego, CA; 3Division of Emergency Medicine, Rady Children’s Hospital, San Diego, CA
Correspondence: Vidyadhar V. Upasani, MD, Department of Orthopedic Surgery, Rady Children’s Hospital, San Diego, 3030 Children’s Way, 3rd Floor, San Diego, CA 92123. E-mail: [email protected]
Received: March 27, 2023; Accepted: June 22, 2023; Published: August 1, 2023
Volume 5, Number 3, August 2023
Abstract
Background: Opioid abuse and overdose are in epidemic range in the United States and medical prescriptions, including those for postoperative analgesia, are a large contributing source to this misuse. Our quality improvement initiative aimed to reduce the opioid prescribing of pediatric orthopaedic surgeons in the postoperative setting. The aim was to decrease the percentage of children with surgically treated supracondylar humerus (SCH) fractures who are prescribed opioid medications at discharge from a baseline of 40% to 10% within 6 months.
Setting/Local Problem: The study took place at an urban level 1 trauma center at a children’s hospital. The orthopaedic team completed closed reduction and percutaneous pinning for SCH fractures over a 14-month baseline period. Forty percent of these patients were discharged with an opioid prescription. After assessing baseline prescription rates, a multidisciplinary team of health professionals developed a key driver diagram.
Interventions: Primary interventions included orthopaedic department-wide pain management education, reporting of prescription rates during monthly conferences, and provider-specific feedback. The primary measure was the percentage of patients prescribed opioids upon discharge following closed reduction and percutaneous pinning of Type II and III SCH fractures. As a balancing measure, we tracked the use of a 24-hour nurse triage line for pain-related follow-up in the intervention period. We used statistical process control to examine changes in measures over time.
Results: The percentage of patients receiving opioid prescriptions upon discharge following surgically treated SCH fractures decreased from 40% to 8% over 5 months and sustained for an additional 16 months.
Conclusions: Through provider education, feedback, and regular reporting, we decreased the number of pediatric patients with surgically treated SCH fractures that were discharged with any opioid prescription by 80% over 5 months while ensuring adequate pain control via postoperative follow-up visits and tracking calls to our nurse triage line.
Introduction
The United States is in the midst of an opioid crisis, contributed to in part by opioids prescribed by orthopaedic surgeons, who are among the highest prescribers amongst physicians. In 2016, they accounted for 5.8% of all opioid prescriptions in the United States, which is the third highest among all specialties.1
A large portion of opioids prescribed by orthopaedic surgeons are prescribed in the postoperative setting.2 While opioids play an important role in postoperative pain control, they can also lead to opioid misuse and addiction in adult and pediatric populations alike. One adolescent study found persistent opioid use in 4.8% of 88,637 13-21 year-olds prescribed opioids postoperatively.3 The annual incidence of opioid-related hospitalization in children 1 to 19 years of age rose 165% between 1997 and 2012, with around 13,052 total hospitalizations due to prescription opioids during this period.4
Many institutions are taking steps to optimize analgesia regimens to limit opioid prescriptions for an array of procedures.5,6 Other groups have focused on demonstrating the efficacy of non-opioid pain medication, with three randomized control trials showing non-opioid pain medication to be as effective as opioids when treating extremity injury pain in children.7–9
Non-opioid pain management following closed reduction and percutaneous pinning (CRPP) for Gartland Type II and III supracondylar humerus (SCH) fractures has become an area of focus within pediatric orthopaedics.10 SCH fractures are the most common elbow fracture in children (age <18), with between 60.3 -71.8 emergency department (ED) visits per 100,000 children annually.11 In a 2012 study of patients treated with CRPP for SCH fracture, there was no significant difference in mean postoperative pain score between patients treated postoperatively with opioids or acetaminophen alone.10 More recently in 2019, a study found that SCH CRPP patients’ pain levels decreased to a clinically unimportant level (<1 on a 10 point scale) by postoperative day three and that patients used <25% of opioid doses prescribed at discharge.12 A subsequent study recommended limiting postoperative oxycodone prescriptions to less than six doses based on finding a mean dosage requirement of four doses.13 Utilizing these findings, one pediatric institution successfully reduced their institution’s opioid prescriptions after CRPP through implementation of a standardized discharge order set.14 They demonstrated a tenfold increase in the number of patients discharged postoperatively without an opioid prescription (4% to 44%).
Given the high volume of these fractures and moderate pain levels associated with CRPP, we identified an opportunity to reduce opioid exposure at our institution. This common procedure also offers an opportunity to build a replicable model for future opioid reduction initiatives in other orthopaedic procedures with similar pain profiles.
Our global aim was to decrease opioid exposure in our pediatric patient population, with the specific objective of reducing the percentage of children with CRPP treated SCH fractures who are prescribed opioid medications upon discharge from a baseline of 40% to 10% over a period of 6 months. Our secondary goal was to reduce opioid prescribing practice variation between senior (>5 years of pediatric orthopaedic practice) and junior pediatric orthopaedic attendings including fellows.
Methods
Context
This quality improvement (QI) study took place at an urban level 1 trauma center at a children’s hospital. In the 2022 fiscal year (July 1, 2021-June 30, 2022), 416 patients presenting to this institution’s ED were diagnosed with SCH fractures. In this same time frame, 269 SCH fractures were managed via CRPP. We included all patients with supracondylar humerus fractures (Gartland Type II and III) that required closed reduction percutaneous pinning. We excluded children that were converted to open reduction and internal fixation as well as children with open fractures, vascular injuries, polytrauma and re-fractures. These 269 patients had an average age of 6.8 years and were 52% male, 52% Hispanic or Latino, 30% white non-Hispanic (NH), 6% Asian NH, 3% Black NH, and 9% other (including Pacific Islander, American Indian or Alaskan native, and other). In our orthopaedic department, all discharge opioid prescriptions are issued by attendings, inclusive of pediatric orthopaedic fellows, and default order sets exist for providers to prescribe non-opioid and/or opioid pain medications upon discharge after surgery. Patients discharged postoperatively have access to a 24-hour nurse triage line and are instructed to return to the emergency department if pain is severe or worsening. This study was granted a QI exemption by our Institutional Review Board.
Planning
The QI initiative was conducted by a multidisciplinary team of health professionals, including pain service, anesthesia, nursing, and orthopaedic surgery. The team assessed baseline prescription rates and provider-specific practices by collecting data from operating room (OR) procedure reports for all cases labeled “Closed Reduction Percutaneous Pinning of Supracondylar Humerus Fracture” within the Electronic Health Record and verified against the Controlled Substance Utilization Review and Evaluation System (CURES). CURES is a database of controlled substance prescriptions dispensed in our state which serves public health agencies and law enforcement. The project team reviewed baseline data over 14 months prior to QI initiation (January 20, 2020-March 21, 2021). The baseline rate of opioid prescriptions after CRPP for SCH fracture was determined to be 40%. During this period, 17 providers prescribed opioids to 86 patients upon discharge. Junior attendings accounted for 61/86 (71%) of total opioid prescriptions, prescribing in 43% of their cases, while senior attendings prescribed in 25% of their cases (p<0.05). A key driver diagram was created from identified key drivers and interventions (Figure 1).
Figure 1. Key driver diagram.
Definitions: SMART, specific, measurable, attainable, relevant, time based; SCH, supracondylar humerus; CRPP, closed reduction percutaneous pinning.

Interventions
Department-Wide Education and Standardization
Best practices for postoperative pain management were determined from literature review and uniform guidelines were presented to attendings at the March 2021 department-wide conference. These guidelines specified that the average, complication-free patient does not require postoperative opioids and should be treated with around-the-clock (ATC) acetaminophen and non-steroidal anti-inflammatory drugs (NSAIDs). In cases which necessitated opioid prescription upon discharge, the recommended prescription for patients under 40 kg was 3 days or 12 doses of 0.05 mg/kg liquid oxycodone to be taken every 6 hours, and patients over 40 kg would receive the same liquid dosing up to 5 mg/dose or 5 mg oxycodone tablets. These dosing recommendations are in agreement with the CDC recommended Michigan Opioid Engagement Network’s Pediatric Prescribing Recommendations.15,16 Our department defined patient-specific exceptions for providers including patients or families with high anxiety about post-discharge pain control, patients with increased inpatient pain requirements, or patients living in a suburban or rural community where liquid opioid medications are less readily available at pharmacies. Continued education of prescribers included presentation of the guidelines at every twice-weekly conference throughout the QI period. Surgeons were encouraged to set expectations with patients and their families that their pain would be adequately controlled without opioids. Patients prescribed opioids were given a standardized hospital handout describing proper storage, use, and disposal of opioids and worrisome signs of opioid overdose.
Steps were taken to mitigate potential undertreatment of pain during this QI initiative. Patients received standard postoperative management, including regular numeric pain rating scale (NPRS) assessments and vital sign monitoring prior to discharge. Providers were educated to verify stable vitals for patients receiving inpatient opioids, evaluate patients with abnormally high NPRS scores, and consider the utility of a discharge opioid prescription. After discharge, children were able to contact their surgical care team via a 24-hour nurse triage call line if evaluation or medication changes were needed prior to regularly scheduled follow-up appointments. These nurse triage calls were recorded and reported to the patient’s respective surgeon during the implementation period. Patients were additionally instructed to return to the institution’s ED, or an ED close to their residence, if pain was severe or worsening.
Group Feedback
Baseline opioid prescription rates between January 2020 and March 2021 were presented anonymously to attendings and fellows at the March 2021 web-based department-wide conference. At each subsequent monthly conference until June 2022, each provider’s non-anonymized monthly opioid prescription rate was presented and compared to both the department-wide rate and the guidelines defined at the March 2021 meeting.
Provider-Specific Feedback
To help decrease practice variation between surgeons and encourage targeted behavior change, provider-specific feedback was given one-on-one in 15-minute meetings, either in person or over video conference to providers with an opioid prescription rate above the department-wide average during the preceding calendar month. The feedback included reviewing the provider’s prescribing rate and how this differed from the guidelines. Other items discussed were patient-specific exceptions including those mentioned in the guidelines and whether non-opioids alone could have been adequate in these cases.
Measures
Primary measures included the percentage of children with surgically treated SCH fractures prescribed opioid medications upon discharge each month. This information was collected using OR reports of all procedures titled “Closed Reduction Percutaneous Pinning Elbow Supracondylar Humerus” within the preceding calendar month. For each procedure reviewed in which opioids were prescribed, the data collected included the medication dosage, medication type, and prescribing provider. Process measures included total opioid prescriptions prescribed per month. Although we did not quantify balancing measures to compare between the pre- and post-implementation periods, there were methods in place to ensure that patient pain was adequately controlled. Specifically, patients were instructed to call the department’s 24-hour nurse triage hotline or return to the ED in the case of severe or worsening pain; these occurrences were tracked during the implementation period alone.
Analysis and Study of Interventions
Discharge opioid prescription data was collected retrospectively on a weekly basis. We used Microsoft Excel (Microsoft Corporation, Redmond, WA) and QI Macros to develop statistical process control charts to define expected variation, examine changes in the mean number of discharge opioid prescriptions over time.17 We used established rules for interpretation of control charts.18 Upper and lower control limits were defined by the distribution of the data, roughly three standard deviations above and below the mean centerline.17 The centerline and upper and lower control limits were revised when introduction of an intervention was associated with special-cause variation as defined by eight consecutive points above or below the mean centerline.
Results
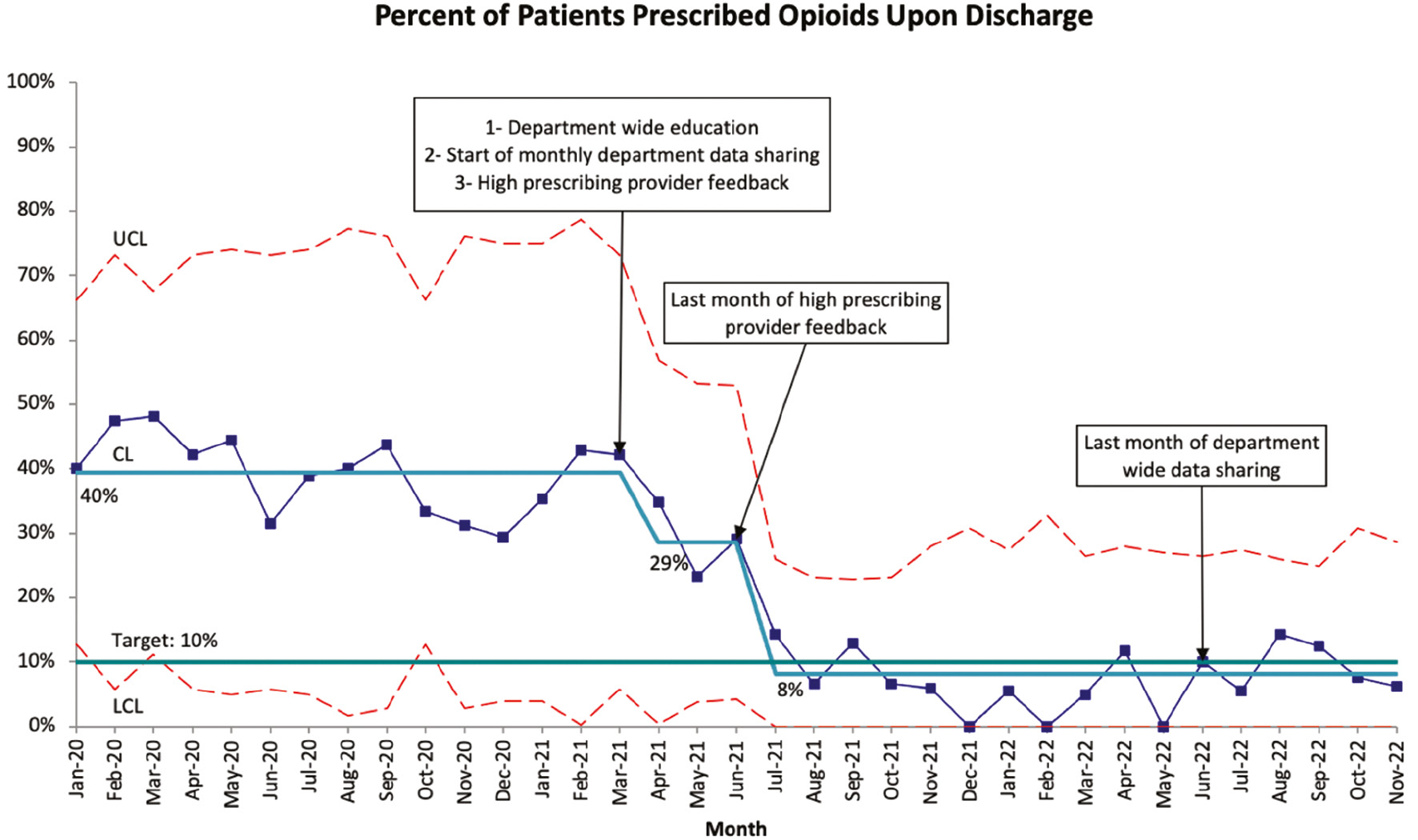
At baseline, 40% of patients that were discharged after CRPP of SCH fracture received an opioid prescription. Five months after the initiative began, this rate had decreased to 8%, surpassing our goal of 10% (Figure 2). We achieved two statistical process control chart shifts associated with our interventions, totaling an 80% reduction in prescribing rate. This change was sustained for an additional 16 months following the time of last shift (July 2021) and 5 months from the time of last intervention (June 2022). Between May 2021 and August 2022, of 359 patients undergoing CRPP for SCH fracture, only two patients contacted the nurse triage line after discharge. One patient was treated in clinic the next day with bivalving of their long arm cast. The second patient was instructed to present to the ED where they received a 3-day supply of liquid oxycodone after evaluation.
Figure 2. Percent of patients each month with surgically treated supracondylar humerus fractures who were prescribed any opioids upon discharge at baseline and after implementation.
Definitions: UCL, upper control limit; LCL, lower control limit; CL, centerline.
Discussion
Our goal was to use QI methodology to reduce opioid prescribing and total opioid exposure of our pediatric orthopaedic patients. Utilizing interventions focused on department-wide education, monthly prescription rate reporting, and targeted provider feedback, we significantly reduced the percentage of children prescribed discharge opioids after surgical treatment of SCH fractures. Further, we found that only two patients contacted our nurse triage line during a 15-month period, and we received positive qualitative feedback from patients, families, and providers during follow-up visits regarding pain levels and satisfaction.
The 80% decrease in the discharge opioid prescription rate was achieved over two process control chart shifts during the implementation period. The first shift in the opioid prescribing rate from 40% to 29% between March and April 2021 was likely driven by project introduction and department-wide education in March 2021. The second shift in the opioid prescribing rate from 29% to 8% between June and July 2021 was potentially associated with targeted providers receiving their second or third round of one-on-one feedback. In total, six providers were given direct feedback, including four junior providers.
We found that targeting high prescribing providers and reviewing case-by-case opioid prescriptions enabled providers to adapt their practices to meet guidelines, which is supportive of several studies that have shown targeted feedback can decrease opioid prescriptions in pediatric ED settings.19–23 We also found that monthly reporting of individual prescribing practices provided competitive motivation and opportunity for discourse on the barriers to implementation. Winslow et al. has demonstrated that peer-comparison feedback could increase the percentage of children discharged after CRPP for SCH fracture with fewer than five opioid doses from 12% to 72%.24 Our QI was able to improve on these results through the addition of the one-on-one feedback, achieving 92% of patients being discharged without opioids.
At baseline, there was a broad distribution of prescribing practices at Rady Children’s Hospital San Diego, with 4/17 providers (23.5%) prescribing opioids in greater than 50% of their cases, 7/17 (41%) prescribing opioids in between 30% and 50% of their cases, and 6/17 (35%) prescribing opioids in less than 30% of their cases. This distribution of prescribing is likely influenced by the providers’ diverse training and pre-conceived notions of postoperative pain requirements. In addition, these practices could have been influenced by the default postoperative order set created previously by our institution’s opioid task force, which allowed for initiation of 3 days of oxycodone. We educated providers that the 3-day threshold does not imply that patients need an opioid prescription, but rather if opioids are prescribed, patients typically do not need more than 3 days’ worth as demonstrated by Nelson et al. and Stillwagon et al.12,13 Otherwise, providers were instructed to follow the proven-equivalent acetaminophen only regimen.10
The baseline practice variation between senior and junior pediatric orthopaedic providers was consistent with Iobst et al. who demonstrated that less experienced providers prescribed significantly higher average opioid doses than experienced physicians.25 This QI was able to reduce practice variation in these groups; during the final 6 months of data collection (June-November 2022), there were 112 surgeries and only 11 discharge opioid prescriptions written. Senior providers wrote opioid prescriptions in 3/43 cases (6.9%) and junior providers in 8/69 cases (11.6%). This 4.7% difference is significantly less than the 18% difference observed at baseline (p<0.05) and replicates results demonstrated by Goodloe et al.14
Practice variation found during this QI could potentially be explained by a lag in adoption of the guidelines by pediatric orthopaedic fellows due to previously engrained practice patterns. Further, the class of fellows starting in June 2021 had not been present at the introduction of the project in March 2021 and were not fully educated on its execution. By July 2021, after additional feedback, this class’s prescription practices mirrored that of the senior providers and were maintained for the duration of the study. Our institution plans to continue our education efforts during onboarding of each new class of fellows as well as the tracking of prescription rates through generation of monthly reports.
The results of this QI could be augmented in the future by changing the default order-set to match the post-implementation practices. This would be supported by Goodloe et al., who demonstrated the success of default analgesia discharge order sets with ibuprofen and acetaminophen.14 Additionally, our institution could standardize administration of intraoperative ketorolac for this procedure, which was found by Adams et al. to be associated with a reduction in postoperative pain, opioid requirements, and length of stay.26
Limitations
Although we only received two calls to our nurse triage line during a 15-month period within the intervention period, our main limitation was our lack of a true balancing measure to quantify pain levels. Further, given our inability to track if patients presented to an outside clinic for pain, our results may underestimate the number of patients with inadequate pain control. Our institution is currently prospectively monitoring postoperative patient pain scores and medication requirements via a text messaging platform, as well as medication-related calls and follow-up appointments.
Inherent to QI projects, the Hawthorne Effect may have influenced providers to decrease their opioid prescribing due to awareness of tracking by the QI team; however, our sustainable low prescription rate, including 5 months beyond the last intervention, mitigate this concern. Additionally, this QI study was performed at a single large-volume pediatric institution and our results may not be applicable to other sites with different patient populations, inpatient care protocols, and/or without access to 24-7 triage care. Further, implementation of this initiative may be less feasible in a patient population with geographical barriers to liquid opioid access. Lastly, this study focused on surgically treated SCH fractures in children, and the results may not apply to all orthopaedic procedures that may require higher intensity analgesia regimens.
Conclusions
In conclusion, the success of this QI initiative provides further support for the implementation of opioid reduction strategies in the postoperative management of SCH fractures. The sustained decrease in prescribing practices reported in our study builds upon the literature, which demonstrates the effectiveness of standardized postoperative pain management protocols and peer-comparison feedback for reducing opioid prescriptions. More globally, this QI provides evidence of successful methodology for addressing one aspect of the opioid epidemic within pediatric orthopaedics. As an institution, we hope to expand this QI to other diagnoses with similar pain profiles, treatments, and postoperative practices. Properly matching analgesia regimens with evidence-based pain requirements will decrease opioid prescription rates while meeting the pain needs of patients.
Acknowledgements
The authors would like to thank Kendall Sanderson, MSOR, MSIHM; Laurel Moyer, MD; and David Pyatt for their significant contributions to the completion of this manuscript.
Additional Links
- Journal of Pediatric Orthopaedics: Current State of the Opioid Epidemic as it Pertains to Pediatric Orthopaedics From the Advocacy Committee of the Pediatric Orthopaedic Society of North America
- POSNAcademy: Comprehensive Pediatric Pain Management Before and After Major Orthopaedic Procedures
- POSNAcademy: Closed Reduction and Percutaneous Pinning Type II Supracondylar Humerus Fracture
- POSNAcademy: Opioids and Children
Disclaimer
No funding was received. The authors report no conflicts of interest related to this manuscript.
References
- Guy GP, Zhang K. Opioid prescribing by specialty and volume in the U.S. Am J Prev Med. 2018;55(5):e153-e155.
- Soffin EM, Waldman SA, Stack RJ, et al. An evidence-based approach to the prescription opioid epidemic in orthopedic surgery. Anesth Analg. 2017;125(5):1704-1713.
- Harbaugh CM, Lee JS, Hu HM, et al. Persistent opioid use among pediatric patients after surgery. Pediatrics. 2018;141(1):e20172439.
- Gaither JR, Leventhal JM, Ryan SA, et al. National trends in hospitalizations for opioid poisonings among children and adolescents, 1997 to 2012. JAMA Pediatr. 2016;170(12):1195-1201.
- Wick EC, Grant MC, Wu CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: a review. JAMA Surg. 2017;152(7):691-697.
- Baker CE, Larson AN, Ubl DS, et al. Tiered guidelines in a pediatric orthopaedic practice reduce opioids prescribed at discharge. J Pediatr Orthop. 2022;42(1):e83-e90.
- Drendel AL, Gorelick MH, Weisman SJ, et al. A randomized clinical trial of ibuprofen versus acetaminophen with codeine for acute pediatric arm fracture pain. Ann Emerg Med. 2009;54(4):553-560.
- Friday JH, Kanegaye JT, McCaslin I, et al. Ibuprofen provides analgesia equivalent to acetaminophen-codeine in the treatment of acute pain in children with extremity injuries: a randomized clinical trial. Acad Emerg Med. 2009;16(8):711-716.
- Poonai N, Bhullar G, Lin K, et al. Oral administration of morphine versus ibuprofen to manage postfracture pain in children: a randomized trial. CMAJ. 2014;186(18):1358-1363.
- Swanson CE, Chang K, Schleyer E, et al. Postoperative pain control after supracondylar humerus fracture fixation. J Pediatr Orthop. 2012;32(5):452-455.
- Holt JB, Glass NA, Shah AS. Understanding the epidemiology of pediatric supracondylar humeral fractures in the United States: identifying opportunities for intervention. J Pediatr Orthop. 2018;38(5):e245-e251.
- Nelson SE, Adams AJ, Buczek MJ, et al. Postoperative pain and opioid use in children with supracondylar humeral fractures: balancing analgesia and opioid stewardship. J Bone Joint Surg Am. 2019;101(2):119-126.
- Stillwagon MR, Feinstein S, Nichols B, et al. Pain control and medication use in children following closed reduction and percutaneous pinning of supracondylar humerus fractures: are we still overprescribing opioids? J Pediatr Orthop. 2020;40(10):543-548.
- Goodloe JB, Bailey EP, Luce LT, et al. A standardized order-set improves variability in opioid discharge prescribing patterns after surgical fixation of pediatric supracondylar humerus fractures. J Surg Educ. 2021;78(5):1660-1665.
- Pediatrics Prescribing Recommendations. OPEN Opioid Prescribing Engagement Network; 2023. https://doi.org/10.56137/OPEN.000055.
- Dowell D, Ragan KR, Jones CM, et al. CDC clinical practice guideline for prescribing opioids for pain — United States, 2022. MMWR Recomm Rep. 2022;71:1-95.
- Brady PW, Tchou MJ, Ambroggio L, et al. Quality improvement feature series article 2: displaying and analyzing quality improvement data. J Pediatric Infect Dis Soc. 2018;7(2):100-103.
- Provost LP, Murray SK. The Health Care Data Guide: Learning from Data for Improvement. John Wiley & Sons; 2022.
- Boyle KL, Cary C, Dizitzer Y, et al. Reduction of opioid prescribing through the sharing of individual physician opioid prescribing practices. Am J Emerg Med. 2019;37(1):118-122.
- Bryl AW, Demartinis N, Etkin M, et al. Reducing opioid doses prescribed from a pediatric emergency department. Pediatrics. 2021;147(4):e20201180.
- Kline TV, Savage RL, Greenslade JH, et al. Affecting emergency department oxycodone discharge prescribing: an educational intervention. Emerg Med Australas EMA. 2019;31(4):580-586.
- Lavingia R, Mondragon E, McFarlane-Johansson N, et al. Improving opioid stewardship in pediatric emergency medicine. Pediatrics. 2021;148(6):e2020039743.
- Kelley-Quon LI, Ourshalimian S, Lee J, et al. Multi-institutional quality improvement project to minimize opioid prescribing in children after appendectomy using NSQIP-pediatric. J Am Coll Surg. 2022;234(3):290-298.
- Winslow L, Holstine J, Samora JB. Reducing the use of opioids for pediatric patients with supracondylar humerus fractures. Jt Comm J Qual Patient Saf. 2020;46(10):581-587.
- Iobst CA, Singh S, Yang JZ. Opioid prescription patterns for pediatric orthopaedic fracture patients. J Clin Orthop Trauma. 2020;11(2):286-290.
- Adams AJ, Buczek MJ, Flynn JM, et al. Perioperative ketorolac for supracondylar humerus fracture in children decreases postoperative pain, opioid usage, hospitalization cost, and length-of-stay. J Pediatr Orthop. 2019;39(6):e447-e451.
