Current Concept Review
Pain After Spine Fusion for Adolescent Idiopathic Scoliosis
Children’s Hospital New Orleans, LSU Health Sciences Center, New Orleans, LA
Correspondence: Manaf H. Younis, MD, MPH, 217 Penn St., Baltimore, MD 21230. E-mail: [email protected]
Received: October 27, 2021; Accepted: March 19, 2022; Published: May 1, 2022
DOI: 10.55275/JPOSNA-2022-0030
Volume 4, Number 2, May 2022
Abstract:
The prevalence and etiology of chronic back pain following posterior spinal fusion with instrumentation (PSF) for adolescent idiopathic scoliosis (AIS) is unknown. We sought to review the prevalence and potential causes of chronic back pain following PSF for AIS. Unfortunately, the definition of chronic pain varies and thus the true prevalence of chronic pain remains unknown but ranges from 16–64.4% 2 years postoperatively. Many patients did not have an obvious etiology of pain identified. Potential causes included mechanical back pain, adjacent segment disease, pseudoarthrosis, implant-related failures, infection, and proximal junctional failure. Common risk factors for these causes of chronic pain include high preoperative pain levels and the degree and type of curve preoperatively.
Key Concepts:
- There is no consensus on the definition of chronic pain after surgery for AIS.
- The prevalence of chronic pain following PSI for AIS ranges from 16-64.4%.
- Mechanical back pain, infection, pseudoarthrosis and implant-related factors are some of the most common causes of chronic back pain following PSI for AIS.
- Systematic work up is needed to arrive at a diagnosis.
Introduction
Adolescent idiopathic scoliosis (AIS) is the most common type of scoliosis. Some reports have shown up to 23% of patients with AIS may have back pain, which is comparable with general populations.1 Surgical correction of spinal deformity may decrease pain in those patients that have pain in the region of the curves. Using the Scoliosis Research Society (SRS) outcome instrument, recent studies have noted an improvement in pain up to 24 months after surgery.2 Yet it is uncertain why some patients may develop pain or continue to have chronic pain following surgery.3 While the true prevalence and etiology of chronic back pain following posterior spinal fusion with instrumentation (PSF) for AIS is unknown, we sought to review the prevalence and potential causes of chronic back pain following PSF for AIS.
Incidence of Chronic Pain
The definition of chronic back pain after PSF for AIS has not been established. Bas et al. examined 104 patients using the SRS-22 questionnaire and found 30.8% of patients reported mild pain, and 6.7% of patients reported severe pain within the last 6 months, but that there was no significant difference in back pain at <2 years, 2–5 years, and >5 years after PSF.4 Bastrom et al. looked at 584 patients with AIS and found that 11% of patients reported pain at least once within 2 years of surgery. Of these patients who reported pain, 21% reported it within 6 months postoperatively and 79% within 6-24 months postoperatively. Within the group that reported pain from 6-24 months postoperatively, 85% of patients had no obvious cause of pain.3 A prospective study of 144 patients undergoing PSF for AIS by Chidambaran et al. found that 37.8% developed “chronic pain” (i.e., 2-3 months postop) and 41.8% developed “persistent pain” (i.e., ≥ 1 year postop).5 The authors found that preoperative pain and higher postoperative opioid needs were significantly predictive of chronic pain—acute pain, chronic pain. Childhood anxiety sensitivity index and surgical duration were also significantly associated with persistent pain. Another study by Helenius et al. examining 55 patients found that at 2 years after surgery, 8 patients had moderate-to-severe pain.2 Landman et al. found that at 1 year after surgery, 68.8% of patients reported mild-to-severe pain within the past 6 months, and 64.4% of patients reported the same findings 2 years postoperatively. Sieberg et al. followed 190 patients for at least 2 years postoperatively, and 77 were followed for at least 5 years postoperatively. After 1, 2, and 5 years, respectively, the authors found that 16%, 16%, and 17% of patients reported moderate-to-severe pain on the SRS-30 questionnaire.4 From these studies it appears that some pain can be intermittently present in up to 65% of patients who undergo PSF for AIS.
Potential Causes of Chronic Pain
Mechanical Back Pain
Mechanical back pain is described as nonspecific pain arising from the spine, intervertebral discs, or surrounding soft tissues. There are many theories about what causes mechanical back pain following spinal surgery. Muscle spasm associated with mechanical back pain may be caused by inflammation of vertebral column tissues; ischemia may also be responsible for causing muscle spasms. Carrilho and Santos suggest that muscle spasms following spinal surgery could be attributed to manipulation of both spinal and peripheral nerves during surgery in the setting of chronic spinal deformity.6 Mechanical back pain following PSF has also been attributed to redistribution of load in spinal segments adjacent to the site of fusion which is more likely to be an issue over the long-term than nerve irritation7. According to Chadbrahman et al., pain following PSF can be a result of shoulder and hip imbalance due to growth and compensation following surgery.5
Mechanical low back pain is a diagnosis of exclusion, so making this diagnosis requires ruling out other causes of back pain, especially those associated with risk of significant complications. In the postoperative population, plain films are important to rule out other sources of pain, especially implant-related problems (Figure 1), progressive deformity, or adjacent degeneration.
Moderate evidence supports the use of NSAIDS and opioids for mechanical back pain in the general population.8 Additionally, a systematic review by Chou et al. reports evidence that non-pharmacologic therapies, such as exercise, physical therapy, spinal manipulation, and massage are also effective in relieving mechanical back pain9.
Figure 1. Pedicle screw fracture with low back pain may indicate pseudarthrosis.

Adjacent Segment Disease (ASD)
ASD is defined by Harrop et al. as “development of clinically symptomatic junctional degeneration.”10 An alternative definition states that ASD involves degeneration of the mobile segments either above or below a fused spinal segment.11 Diagnosis involves radiographic evaluation with x-ray, CT, and MRI. Some parameters commonly used to diagnose ASD in the adult PSF population include the development of spondylolisthesis >3-4 mm, retrolisthesis >4 mm, a decrease in disk height by more than 3 mm or 10%, complete collapse of the disk space, angle change >10 degrees between adjacent vertebral bodies on flexion and extension radiographs, segmental kyphosis >10 degrees, intervertebral angle at flexion <-5 degrees, hypertrophic facet joint arthropathy, osteophyte >3 mm, and compression fracture.12
Although the percentage of patients who develop ASD following PSF varies among studies, one study that specifically addressed adolescents found that 16% of patients who had normal discs on initial preoperative imaging developed ASD.13 Most cases of ASD develop within the first 3-5 years following initial PSF.13
Studies have shown that anywhere from 2.6-27.4% of adult patients who have been treated with lumbar PSF undergo additional surgery for ASD that did not respond to conservative treatment.12 This number is likely lower in the short term among AIS patients who tend to be younger and, therefore, have less degeneration; however, in the long term, it may prove higher as the fusion constructs are longer and the patients have more years of life ahead of them to develop degeneration. Treatment of ASD in AIS is typically based on symptoms or the severity of the developing deformity; management involves extending the fusion to incorporate the affected area to prevent further degeneration and deformity.
Pseudoarthrosis
Although uncommon with modern techniques, pseudoarthrosis is another potential source of pain following PSF. Pseudoarthrosis has been defined as “the absence of bony fusion 1 year” post PSF.14 Pseudoarthrosis typically manifests months to years after initial PSF as axial or radicular pain or implant failure (broken or loosened implants) but can also be asymptomatic (Figure 2). The reported incidence of pseudoarthrosis varies among studies. One systematic review involving 16,938 pediatric patients who underwent spinal surgery for AIS reported a 1.4% occurrence of pseudoarthrosis following spinal deformity surgery.15 Overall, some risk factors for pseudoarthrosis include insufficient or poor-quality bone grafting, inadequate stability of the fusion construct, and smoking. Imaging studies, such as x-ray and CT are used in diagnosis but are less sensitive than direct surgical exploration. Several studies including one by Quon et al. advocate for the use of PET imaging over CT imaging in diagnosing nonunion following spinal fusion.16
Figure 2. Two years after PSF this patient had worsening in her chronic back pain. The radiographs demonstrated rod fracture and CT confirmed pseudarthrosis.
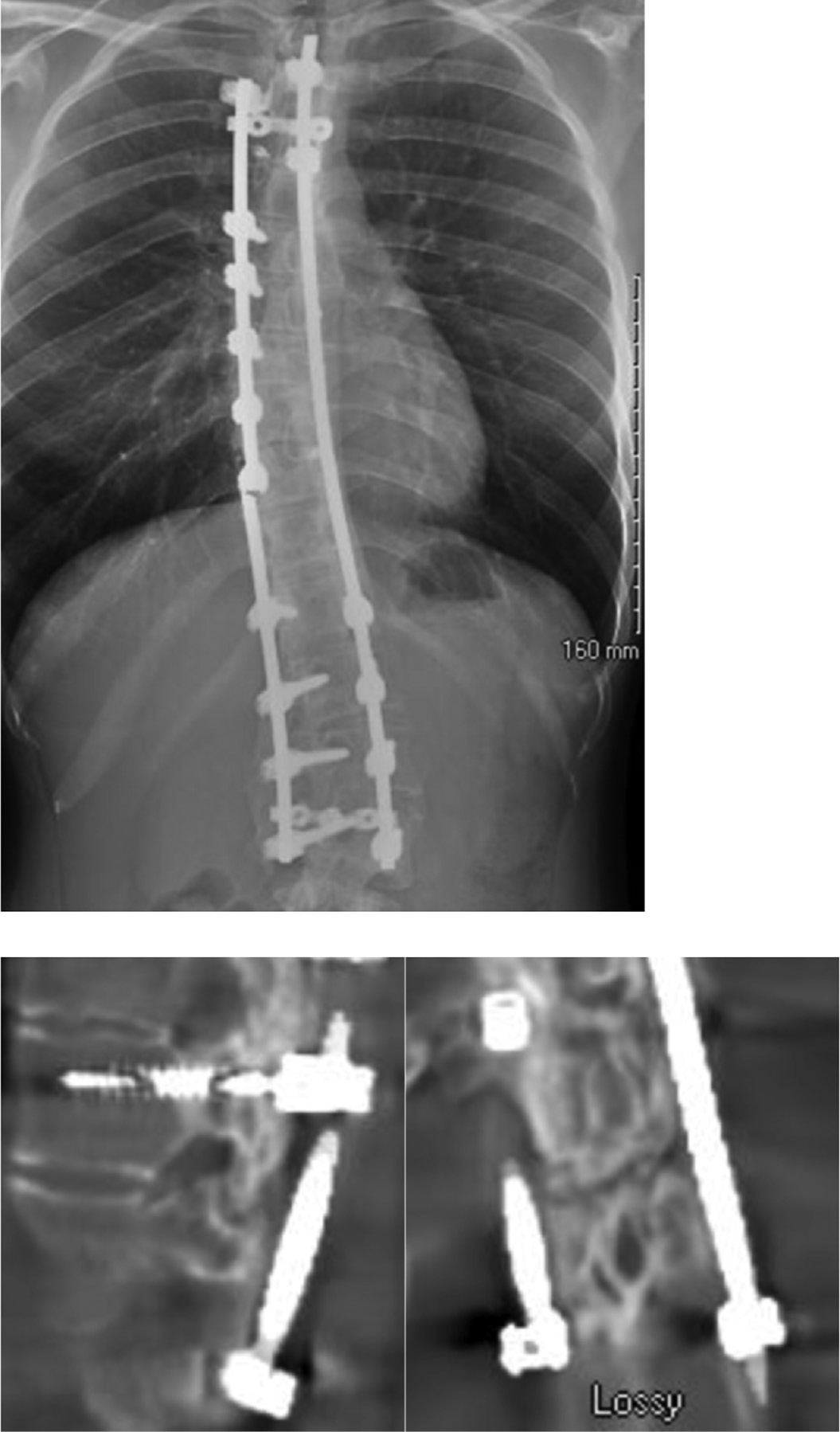
If conservative treatment fails to improve pain in symptomatic patients, attempts at a second fusion are typically made. The decision to perform additional surgery is usually determined by the degree and persistence of the pain. Rates of successful fusion also depend on addressing the underlying issues that contributed to the pseudoarthrosis.
Implant-Related Factors: Failure/ Malposition/Prominence
A review of pedicle screw complications in pediatric scoliosis surgery found that 4.2% of screws were mal-positioned (Figure 3), based on any breach of the pedicle. However, in studies where CT scans were obtained on all patients, the rate of screw malposition increased to 15.7%. Among the studies reviewed, the reported rate of revision surgery for mal-positioned screws was only 0.83%.17
Figure 3. In addition to more severe risks, the mal-positioned screw on the left may be a pain generator. The patient on the right had canal compromise that led to 9 months of severe neuritic pain.

In a study of 120 patients undergoing PSF for AIS, pedicle screw loosening was more likely at the upper instrumented vertebra (UIV) or the lowest instrumented vertebra (LIV) compared to other vertebrae.18 Mal-positioned screws can cause pain perhaps due to direct contact with neural elements or inadequate fixation leading to excess motion.
Infection
Deep surgical site infections (SSIs) are a potential cause of pain following PSF for AIS. This should be considered in the patient who has chronic non-specific back pain that may or may not have redness or drainage but may have slight elevations of inflammatory parameters such as the ESR. A retrospective review of 1071 patients found an overall incidence of 3.6% with an increased incidence of 8.3% following revision surgery.19 Other reports have found an incidence between 1.4% and 6.9%.20 Incidence of infection is lower in AIS compared to neuromuscular, congenital, and syndromic scoliosis.19 These infections are thought to occur through either direct seeding at the initial operation followed by a latent period and re-activation or by later hematogenous spread. A retrospective review of 15 patients that developed a late SSI following PSF for AIS found the mean time to infection was 70 months.21 The most common organisms associated with late SSIs include Staphylococcus epidermidis, Staphylococcus aureus, Propionibacterum acnes, Serratia marcescens, and coagulase negative staph.19
Specific risk factors for delayed SSI after PSF for AIS have not been well-established but do include significant past medical history, increased drainage when a drain was used, no use of a drain postoperatively, extension of the fusion level distally, blood transfusion, increased return of cell saver blood intraoperatively, and use of stainless steel alloy implants compared to titanium implants for delated SSIs.21
While no treatment algorithm has been universally accepted, antibiotics with irrigation and debridement is the most common treatment choice regardless of time from the index procedure. For patients who develop early infections (within 90 days of the index procedure), the goal should be to retain PSF hardware. Antibiotic therapy should be tailored to cultures obtained intraoperatively and will often require at least 6 weeks of intravenous therapy. There is no good evidence to guide when implant removal for persistent infection should be pursued. The fusion mass must be evaluated, typically by CT scan, to consider the risk of curve progression following implant removal. Implant removal may be complicated by progression of deformity.22 The long-term follow-up of a cohort of 21 patients showed a “settling” effect in the coronal plane of the main thoracic and TL/L curves after instrumentation removal.23 Muschik et al. reviewed 45 patients who underwent PSF for scoliosis and experienced development of late infections and after a mean of 3 years after the initial procedure, either underwent implant removal alone or additionally underwent re-instrumentation and fusion. At follow-up, the outcome was clearly better in re-instrumented patients.24
Proximal Junctional Kyphosis (PJK) and Proximal Junctional Failure (PJF)
PJK is a sagittal plane deformity that can develop after spinal deformity correction. PJF has recently been defined as symptomatic PJK requiring revision surgery.25 Lee et al. defined PJK as ≥5 degrees kyphosis greater than normal from T2 to the most proximal level of instrumented fusion in patients with AIS.26 Although it is not uncommon for patients to develop radiographic evidence of PJK following PSF for AIS, the clinical significance of these findings does not always correlate with radiographs (Figure 4). Kim et al. found no significant differences in pain and self-image SRS subscores between patients who developed PJK and those who did not following PSF for AIS.27 Since there are multiple ways to define PJK, the prevalence in patients with AIS varies in the literature. Most reports suggest that anywhere from 9-46% of AIS patients who undergo PSF develop PJK.28
Figure 4. This female with AIS underwent PSF and developed painful PJK.

Many approaches have been suggested to reduce PJK incidence including minimizing paraspinal dissection around the UIV, avoiding disruption of the supraspinous and interspinous ligaments, proper end vertebra selection, “soft landing” at UIV, and restoring sagittal balance.28 Multiple risk factors have been linked to PJK including thoracoplasty, preoperative thoracic hyperkyphosis, hybrid instrumentation, combined anterior-posterior spinal fusion, and fusion to the lower lumbar spine and sacrum.28
Development of severe symptoms or deformity is often used as the benchmark for intervention, and treatment typically involves extending the fusion construct proximally to prevent further deformity.
Metal Allergy
A local immune reaction to the metal implants used in PSF has been a potential cause of postoperative pain. Metal hypersensitivity following PSF can present as pain at the surgical site, swelling, skin reactions, or radiculopathy making it difficult to differentiate from other postoperative causes of pain. Metal hypersensitivity is thought to involve a local cell-mediated reaction to metal ions that are likely released from the implant surfaces over time and can lead to aseptic inflammation and possible loosening of the implants.29 Metal hypersensitivity is a delayed Type IV hypersensitivity reaction that gradually affects the surrounding tissues of the implan.29 Given the gradual nature of the immune reaction, patients with a metal allergy to the implant typically experience pain-free interval postoperatively with symptom onset several months after the initial surgery.30
Metal hypersensitivity in patients with postoperative pain who underwent PSF is another diagnosis of exclusion. Overall, metal hypersensitivity should be considered as a rare cause of pain after PSF in cases where symptoms are not explained by other mechanisms such as infection and implant failure. Metal hypersensitivity can cause implant loosening and corrosion which both can be identified on CT in some cases.29 Biopsy is considered the most accurate detector of metal allergy: a predominantly lymphocytic infiltrate suggests hypersensitivity while a neutrophilic infiltrate suggests infection.31 The current literature on this condition recommends hardware removal as the only curative intervention.32
Authors’ Preferred Workup Algorithm and Management
Overall, there are no definitive guidelines on timeline of the diagnostic workup. Our initial approach is to review a detailed history focusing on the course of pain and possibility of psychological causes, including addiction to pain medications. A thorough review of symptoms including items such as night sweats, fevers, and chills could indicate possible infection. Physical exam should include gait assessment, ROM, tenderness, and neurologic exam. Evaluation of the wound for any redness, swelling sinus tracts, or dehiscence is necessary. Physical examination should also include assessing Waddell signs, which include superficial or non-anatomic pain on palpation, pain during painless evaluation, or overreaction to stimuli which can indicate a psychological component. Exam should also evaluate for sacroiliac or hip pathology that can mimic back pain.
Standing AP, lateral, and flexion-extension plain radiographs to assess alignment and implant position are needed to rule out obvious issues. We have a low threshold for infectious workup with ESR, CRP, and WBC. If there is a low-grade elevation in these parameters, a careful history is taken to include any recent illnesses. Often times these tests need to be repeated in a month to demonstrate persistent elevation, which raises the index of suspicion for a nascent infection.
Advanced imaging can be ordered to evaluate for pain generators such as pseudarthrosis, implant failure or malposition, adjacent segment disease, or infection. An MRI with and without gadolinium enhancement can demonstrate a variety of pathologies, such as signs of infection and adjacent segment disease. CT scans are used to assess accurate positioning of implants, bone union, and rule out pseudoarthrosis. Whether to do a CT scan or MRI is guided by presentation and differential diagnosis: MRI if concerned for infection or degenerative disc disease versus CT for mal-positioned implants or pseudoarthrosis. As a result of image artifact, it can be hard to detect infection on advanced imaging. If infection is strongly suspected, ultrasound can detect fluid collections and guide aspiration and culture.
For the patient who is at least a year from PSF and has unremitting pain with negative x-rays, blood work, and whose CT scan demonstrates solid fusion, we consider exploration, clinical assessment of bone union and possible implant removal. Shared decision-making is needed to take this step as some patients can continue to have pain and possible curve gradual curve progression (Figure 5).
Figure 5. This 14-year-old with AIS and a history of mild back pain and anxiety undergoes PSF for progressive AIS. She had persistent mid back pain for 5 years despite conservative management. Blood work and CT scan were negative for infection and implant or pseudarthrosis. At age 20 she undergoes implant removal and has had a decrease in her pain at most recent follow-up.

Summary
After a thorough literature review, the true prevalence of chronic pain following surgery remains difficult to quantify. Each study defines “chronic pain” in the context of PSF in their own terms. The true prevalence of chronic pain remains unknown but ranged in previous studies from 16-64.4% 2 years postoperatively. Common risk factors for these causes of chronic pain include high pre-operative pain levels and the degree and type of curve preoperatively. Many patients do not have an obvious etiology of pain and causes include mechanical back pain, adjacent segment disease, pseudoarthrosis, implant-related failures, infection, and proximal junctional failure which require a thorough investigative work-up for diagnosis.
Additional Links
- POSNAcademy: Anterior Lumbar Vertebral Body Tethering in Adolescent Idiopathic Scoliosis, Courtney E. Baker, MD; Todd A. Milbrandt, MD; D. Dean Potter, MD; A. Noelle Larson, MD—https://bit.ly/3u78b2Q
- Webinar: Best Practices for Pediatric, Spine, and Back Pain, Columbia Orthopedics—https://bit.ly/38wfIQB
- OrthoKids: Back Pain in Children—https://bit.ly/3DSyugF
Disclaimer
No funding was obtained for this study. The authors have no conflicts of interest to disclose.
References
- Asher MA, Burton DC. Adolescent idiopathic scoliosis: natural history and long term treatment effects. Scoliosis. 2006;1(1):2.
- Helenius L, Diarbakerli E, Grauers A, et al. Back pain and quality of life after surgical treatment for adolescent idiopathic scoliosis at 5-year follow-up: comparison with healthy controls and patients with untreated idiopathic scoliosis. J Bone Joint Surg AM. 2019;101(16):1460-1466.
- Bastrom TP, Marks MC, Yaszay B, et al. Prevalence of postoperative pain in adolescent idiopathic scoliosis and the association with preoperative pain. Spine (Phila Pa 1976). 2013;38(21):1848-1852.
- Sieberg CB, Simons LE, Edelstein MR, et al. Pain prevalence and trajectories following pediatric spinal fusion surgery. JPain. 2013;14(12):1694-1702.
- Chidambaran V, Ding L, Moore DL, et al. Predicting the pain continuum after adolescent idiopathic scoliosis surgery: a prospective cohort study. Eur J Pain. 2017;21(7):1252-1265.
- Carrilho PEM, Dos Santos MBM. Focal muscle spasms after thoracic spine surgery for schwannoma: the twitching scar. J Mov Disord. 2020;13(2):168–170.
- Rigoard P, Blond S, David R, et al. Pathophysiological characterisation of back pain generators in failed back surgery syndrome (part B). Neurochirurgie. 2015;61(Suppl 1):S35-S44.
- Will JS, Bury DC, Miller JA. Mechanical low back pain. Am Fam Physician. 2018;98(7):421-428.
- Chou R, Deyo R, Friedly J, et al. Nonpharmacologic therapies for low back pain: a systematic review for an American College of Physicians clinical practice guideline. Ann Intern Med. 2017;166(7):493-505.
- Lee CS, Hwang CJ, Lee SW, et al. Risk factors for adjacent segment disease after lumbar fusion. Eur Spine J. 2009;18(11):1637-1643.
- Hoogendoorn RJ, Helder MN, Wuisman PI, et al. Adjacent segment degeneration: observations in a goat spinal fusion study. Spine (Phila Pa 1976). 2008;33(12):1337-1343.
- Charles Malveaux WMS, Sharan AD. Adjacent segment disease after lumbar spinal fusion: a systematic review of the current literature. Semin Spine Surg. 2011;23(4):266-274.
- Ghandhari H, Ameri E, Nikouei F, et al. Long-term outcome of posterior spinal fusion for the correction of adolescent idiopathic scoliosis. Scoliosis Spinal Disord. 2018;13:14.
- Peters M, Willems P, Weijers R, et al. Pseudarthrosis after lumbar spinal fusion: the role of 18F-fluoride PET/CT. Eur J Nucl Med Mol Imaging. 2015;42(12):1891-1898.
- How NE, Street JT, Dvorak MF, et al. Pseudarthrosis in adult and pediatric spinal deformity surgery: a systematic review of the literature and meta-analysis of incidence, characteristics, and risk factors. Neurosurg Rev. 2019;42(2):319-336.
- Quon A, Dodd R, Iagaru A, et al. Initial investigation of 18F-NaF PET/CT for identification of vertebral sites amenable to surgical revision after spinal fusion surgery. Eur J Nucl Med Mol Imaging. 2012;39(11):1737-1744.
- Hicks JM, Singla A, Shen FH, et al. Complications of pedicle screw fixation in scoliosis surgery: a systematic review. Spine (Phila Pa 1976). 2010;35(11):E465-E470.
- Uehara M, Takahashi J, Ikegami S, et al. Pedicle screw loosening after posterior spinal fusion for adolescent idiopathic scoliosis in upper and lower instrumented vertebrae having major perforation. Spine (Phila Pa 1976). 2017;42(24):1895-1900.
- Warner SJ, Uppstrom TJ, Miller AO, et al. Epidemiology of deep surgical site infections after pediatric spinal fusion surgery. Spine (Phila Pa 1976). 2017;42(3):E163-E168.
- Coe JD, Arlet V, Donaldson W, et al. Complications in spinal fusion for adolescent idiopathic scoliosis in the new millennium. A report of the Scoliosis Research Society Morbidity and Mortality Committee. Spine (Phila Pa 1976). 2006;31(3):345-349.
- Di Silvestre M, Bakaloudis G, Lolli F, et al. Late-developing infection following posterior fusion for adolescent idiopathic scoliosis. Eur Spine J. 2011;20(Suppl 1):S121-S127.
- Rathjen K, Wood M, McClung A, et al. Clinical and radiographic results after implant removal in idiopathic scoliosis. Spine (Phila Pa 1976). 2007;32(20):2184-2188.
- Potter BK, Kirk KL, Shah SA, et al. Loss of coronal correction following instrumentation removal in adolescent idiopathic scoliosis. Spine (Phila Pa 1976). 2006;31(1):67-72.
- Muschik M, Luck W, Schlenzka D. Implant removal for late-developing infection after instrumented posterior spinal fusion for scoliosis: reinstrumentation reduces loss of correction. A retrospective analysis of 45 cases. Eur Spine J. 2004;13(7):645-651.
- Yagi M, Rahm M, Gaines R, et al. Characterization and surgical outcomes of proximal junctional failure in surgically treated patients with adult spinal deformity. Spine (Phila Pa 1976). 2014;39(10):E607-E614.
- Lee GA, Betz RR, Clements DH, 3rd, et al. Proximal kyphosis after posterior spinal fusion in patients with idiopathic scoliosis. Spine (Phila Pa 1976). 1999;24(8):795-799.
- Kim YJ, Bridwell KH, Lenke LG, et al. Proximal junctional kyphosis in adolescent idiopathic scoliosis following segmental posterior spinal instrumentation and fusion: minimum 5-year follow-up. Spine (Phila Pa 1976). 2005;30(18):2045-2050.
- Lee J, Park YS. Proximal junctional kyphosis: diagnosis, pathogenesis, and treatment. Asian Spine J. 2016;10(3):593-600.
- Shang X, Wang L, Kou D, et al. Metal hypersensitivity in patient with posterior lumbar spine fusion: a case report and its literature review. BMC Musculoskelet Disord. 2014;15:314.
- Guyer RD, Shellock J, MacLennan B, et al. Early failure of metal-on-metal artificial disc prostheses associated with lymphocytic reaction: diagnosis and treatment experience in four cases. Spine (Phila Pa 1976). 2011;36(7):E492-E497.
- Davies AP, Willert HG, Campbell PA, et al. An unusual lymphocytic perivascular infiltration in tissues around contemporary metal-on-metal joint replacements. J Bone Joint Surg Am. 2005;87(1):18-27.
- Zairi F, Remacle JM, Allaoui M, et al. Delayed hypersensitivity reaction caused by metal-on-metal total disc replacement. J Neurosurg Spine. 2013;19(3):389-391.
